Lehrbuch Lyme-Borreliose
4 |
Infectiologic differential diagnosis of chronic Lyme disease and so-called coinfections |
Table of contents
Yersinia enterocolitica infection (yersiniosis)
Mycoplasma pneumoniae infection
Secondary coinfections of Lyme disease
Human granulocytic anaplasmosis (HGA)
Human parvovirus B19 infection
Overview of the symptomatology and treatment of LD and chronic coinfections
Summary
In cases of Lyme disease (LD), other infections can be concurrently present; their pathological synergism exacerbates the disease state or induces the same disease manifestations. Such
concomitant infections are termed coinfections. The coinfections can also be transmitted by ticks as LD is, i.e. a tick bite can result in multiple infections. A fraction of the coinfections is
transmitted independently of ticks or in addition to tick transmission there are other modes of transmission. The clinically relevant coinfections are caused by Bartonella species, Yersinia
enterocolitica, Chlamydophila pneumoniae, Chlamydia trachomatis, and Mycoplasma pneumoniae. In contrast to the USA, human granulocytic anaplasmosis (HGA, previously called human granulocytic
ehrlichiosis (HGE)) and babesiosis are not of major importance in Europe. The “coinfections” can, of course, also occur without connection to the LD and in some cases independently result in
pronounced disease symptoms, which exhibit substantial instances of overlap with LD’s clinical picture. This applies particularly to infections caused by Bartonella henselae, Yersinia
enterocolitica, and Mycoplasma pneumoniae. Chlamydia trachomatis primarily results in arthritides; Chlamydophila pneumoniae additionally causes disease manifestations of the nervous system and of
the heart. This makes differential diagnosis very difficult; in some cases, impossible. Even more problematical is the diagnosis situation when coinfections occur in association with LD, i.e.
when double and multiple infections exist. — The pathological importance of the coinfections was first recognized in the 1990s, i.e. approximately ten years after the discovery of LD. No studies
exist on the treatment of coinfections; the therapy recommendations are based on individual expert opinions. In antibiotic treatment, the use of 3rd generation cephalosporins should only be
considered in cases of Lyme disease. The same applies to carbapenem, which is also occasionally used in infections caused by Yersinia enterocolitica subsequent to testing. For the remaining
infections tetracyclines and macrolides are predominantly used; quinolones are an alternative, particularly gemifloxacin. For Bartonella henselae, Chlamydia trachomatis, and Chlamydophila
pneumoniae the combination with rifampicin is recommended. Erythromycin is the pharmaceutical agent of choice for Campylobacter. The symptomatology and antibiotic treatment of the infectious
diseases are presented in tabular overviews at the end of the text.
In cases of Lyme disease (LD), other infections, whose pathological synergism exacerbate the disease state or induce the same disease manifestations, can exist concurrently. Such concomitant
infections are termed coinfections. The coinfections can also be transmitted by ticks as LD is, i.e. a tick bite can result in multiple infections. A fraction of the coinfections is transmitted
independently of ticks, or in addition to tick transmission there are other modes of transmission.
The coinfections that are transmitted by ticks are given in Table 4.1; the coinfections that are independent of ticks are compiled in Table 4.2.
In Europe ticks are not only infected by Borrelia burgdorferi but also by other pathogens i.e. Human Granulocytic Anaplasmosis, Rickettsia, Coxiella burnetii, Babesia microti and Babesia divergens [351-354].
The coinfections favor the expression of disease states by means of immune system modulation and are considered to be a major reason for therapy resistances [176 - 192].
The importance of the coinfections for the pathological process—i.e. their pathogenicity compared to Lyme disease—has not been clarified. Thus, in cases of double or multiple infections, a decision cannot be made as to which infection dominates in the pathological process.
In the symptomatology there are substantial overlaps between Lyme disease and the coinfections so that an unequivocal assignment of the disease manifestations to the existing infections is impossible. Many symptoms can thus be due to both a Lyme disease and the so-called coinfections.
The problematic nature of Lyme disease and coinfections always concerns the chronic course. The coinfections are thus only of importance for chronic Lyme disease (late stage, stage III). On the other hand, the synergic-pathological mechanism requires that the coinfections also are present in a chronic persistent form.
Anamnistically, one has to consider whether coinfections occurred in their acute form in the early phase because this contributes to the recognition of coinfections in the chronic phase.
In terms of laboratory diagnostic tests, only methods for indirect pathogen detection (serology, LTT) are also available for the coinfections in most cases, as is the case for Lyme disease. The prior infection can be confirmed with serological investigations. However, a positive serological finding is not proof that the infection caused the current illness. Basically, it is neither possible to prove the presence of an infectious disease nor to exclude it by means of a serological finding. Only if pathological laboratory findings occur or a deterioration of the finding can be detected in correlation with the disease are conclusions as to the disease development and situation justified to a certain degree in cases of previous seronegitivity or negative LTT in temporal parallelism to disease development.


The significant coinfections of Lyme disease are caused by Bartonella species (primarily by B. henselae), Chlamydia trachomatis, Chlamydophila pneumoniae, Yersinia enterocolitica, and Mycoplasma
pneumoniae. Accordingly, these infectious diseases are highlighted in Tables 4.1 and 4.2 by the typeface.
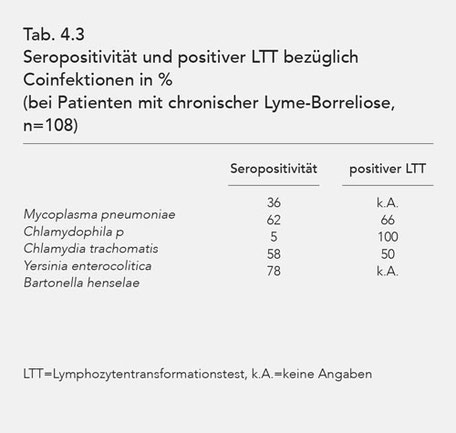
The frequency of seropositivity and positive LTT (lymphocyte transformation test) was studied on my own clientele (n = 108). The results are presented in Table 3. The investigations were
conducted at the Institute for Medical Diagnostics (IMD) in Berlin. For more detailed information, please refer to the corresponding reference [339]. An LTT is not available for Bartonella. An
LTT was not performed for Mycoplasma pneumoniae. The high incidence of the Chlamydia trachomatis LTT is conspicuous and unresolved.
According to investigations on my own clientele, CD57 NK cells are frequently diminished in chronic Lyme disease, but seldom in cases involving coinfections. However, the basic principle is that CD57 NK cells can be diminished in all chronic infectious diseases, but the phenomenon is observed relatively frequently in chronic LD.
The more important coinfections are summarized in an overview (Table 4). These more important coinfections are presented in accordance with the tabular sequence; they are then followed by the less important ones (chapter “Secondary coinfections of Lyme disease”) and finally to round out the presentation, a chapter on the so- called reactive arthritis follows.
In contrast to the USA, HGA (human granulocytic anaplasmosis, syn. human granulocytic ehrlichiosis (HGE)) and babesiosis are of little importance as coinfections in Europe.

Bartonellosis
Many interrelationships in the bartonellosis’ mode of transmission have not yet been clarified. The important infection data described in the scientific literature are summarized in Table 4.5.
Primary manifestations: infected skin lesion, swollen lymph nodes, multiorgan disorder (e.g. liver, spleen, nervous system, eye) [66-67], cf. Tab. 4.6.
Until 1993 only B. bacilliformis was known. The different Bartonella subspecies were first described [69] and their pathological significance, recognized in 1993 Bartonellosis can be expected to have substantial significance as a Lyme disease coinfection. With regard to the health policy aspect, Lyme disease is more important because of its frequency. However, in this context it should be noted that bartonellosis has not been nearly as intensively investigated as Lyme disease. Additionally, it is obvious from my own observations that the serology for Bartonella is frequently positive in patients with chronic Lyme disease.
With the increasing development of laboratory tests which is to be expected, the currently underestimated prevalence of bartonellosis will be more correctly registered in the future, and the importance of this disease will also be determined on the basis of its frequency.
Bartonellosis (caused by B. henselae und B. bacilliformis) can be associated with an extraordinary variety of symptoms. For further information on this see Table 4.8.
The bacterial-inflammatory skin infection (scratch or bite location) is in no way obligatory, i.e. bartonellosis can also occur without the typical cat scratch disease, which is characterized by the infected skin lesion and lymph node swelling.
Bartonellosis’ disease morphology can be better recognized when the most important of its many symptoms (Table 4.8), i.e. its main manifestations, are considered (cf. Table 4.6).
There are numerous overlaps with Lyme disease in the disease manifestation of bartonellosis. This fact is also mentioned in the relevant current literature [86].
The laboratory diagnostics for bartonellosis is given in Table 4.7.




In cases of infection with Bartonella, the blood smear initially shows pathogens on the outer membrane; in the further course the pathogens are increasingly localized intracellularly. In the process, the light colored center of the erythrocytes is lost (Fig. 1).
There is no information on the value of serology in the literature. In particular, the question as to whether seronegativity rules out the disease has not been clarified. On the other hand, as is the case for many other infectious diseases, a positive serological finding merely proves that an infection took place, but does not indicate that the disease currently exists.
Detecting Bartonella in culture is extremely problematical, and the sensitivity is very low so that this method of investigation is not part of routine diagnostics.

Detection of pathogens using PCR in biopsies appears to be very promising [87, 232], but the investigation with PCR must follow the biopsy nearly immediately [88].
The chronic course of bartonellosis has been described in numerous studies, in part, on large groups [90 - 94]. The long duration of the disease, which frequently covers many years, and the high similarity of the disease manifestations makes it extremely difficult to distinguish from chronic Lyme disease. Thus, bartonellosis is of great importance in the infectiologic differential diagnosis of Lyme disease.
In this context, attention should be paid to the fact that B. henselae has been found in ticks and that the transmission of B. henselae by ticks has been documented by means of pathogen detection in the liquor [95]. Additionally, the prevalence of B. henselae in ticks is apparently high; scientific studies determined a prevalence of 40% [96].
According to my own surveys, 78% of the patients with chronic Lyme disease proved to be seropositive for Bartonella henselae.
A particular characteristic of Bartonella is the induction of vascularized tumors or granulomas, which occur in the region of the skin (bacillary angiomatosa), in the liver (peliosis hepatis) or in the spleen (peliosis splenitis) [97 - 99]. In this context, these vascular tumors or granulomas exhibit a pathological sprouting of capillaries as well as enlarged and hyperproliferative vascular endothelial cells [100]. Bartonellosis is obviously accompanied by a stimulation of blood vessel formation. This corresponds to the observation that bartonellosis, in addition to angiomatosis, also results in different other skin manifestations in which an increased vessel formation is observed [101]. In addition, the determination of VEGF (vascular endothelial growth factor) in blood could be of diagnostic importance [101].
In all pathogenetic relevant bartonellae (B. quintana, B. henselae, B. bacilliformis), this effect on endothelial cells and the induction of angiogenesis has been demonstrated. In the process, the vascular proliferation was primarily traced back to three factors [102 - 108]:
- Elevated endothelial cell proliferation
- Inhibition of the apoptosis of endothelial cells
- Increased secretion of vasculoproliferative cytokines
All these studies support the idea that VEGF (vascular endothelial growth factor) plays a significant role in Bartonella-induced endothelial cell proliferation [108].
Bartonellae are localized in erythrocytes and cause deformations of the erythrocyte membrane [110 - 111].
The visualization of the bartonellae in erythrocytes is already being used diagnostically, particularly also with regard to the extent of the infection [101]. However, irrefutable literature on erythrocyte infestation in chronic bartonelloses does not exist. The diagnostic value of a new method of detecting pathogens by means of cilia in blood smears cannot be assessed yet.
The formation of intracellular blebs subsequent to penetration into the endothelial cells has also been determined for Bartonella quintana, i.e. a similar process to that in Lyme disease. Moreover, Borrelia burgdorferi also has a high affinity for endothelial cells, and the development of blebs, particularly in chronic disorders, has been described. In connection with Lyme disease, the intracellular presence of the pathogen and the formation of biologically less active eukaryotic forms (cyst forms, blebs) have been discussed as the cause of the failure of antibiotic treatment. In addition, parallels between Lyme disease and bartonellosis have been found [cf. 112 - 117].
For completeness sake, two additional forms of bartonellosis are mentioned, namely Oroya fever or verruga peruana and trench fever.
Oroya fever and Verruga peruana are a Bartonella bacilliformis infection, which is transmitted by sand flies. The disease occurs in the Andes. The acute form affects tourists who are immunologically naive with respect to B. bacilliformis. Without treatment the mortality rate is 40%. To date, the factor which results in the severe disease course in this context is unknown.
Oroya fever and its recognition as an infectious disease date back to Carrion, who verified that the disease is infectious in a fatal self-test at the end of the 19th century.
Trench fever was discovered at the beginning of the 20th century. Its transmission occurs via lice [127 - 129]. In 2002 the pathogen was detected for the first time in erythrocytes; as a result the transmission by lice became plausible [130].
No adequate studies exist for the treatment of bartonellosis. There is not a single treatment method that has been approved by the FDA, CDC or IDSA [101]. This applies particularly for chronic courses [123].
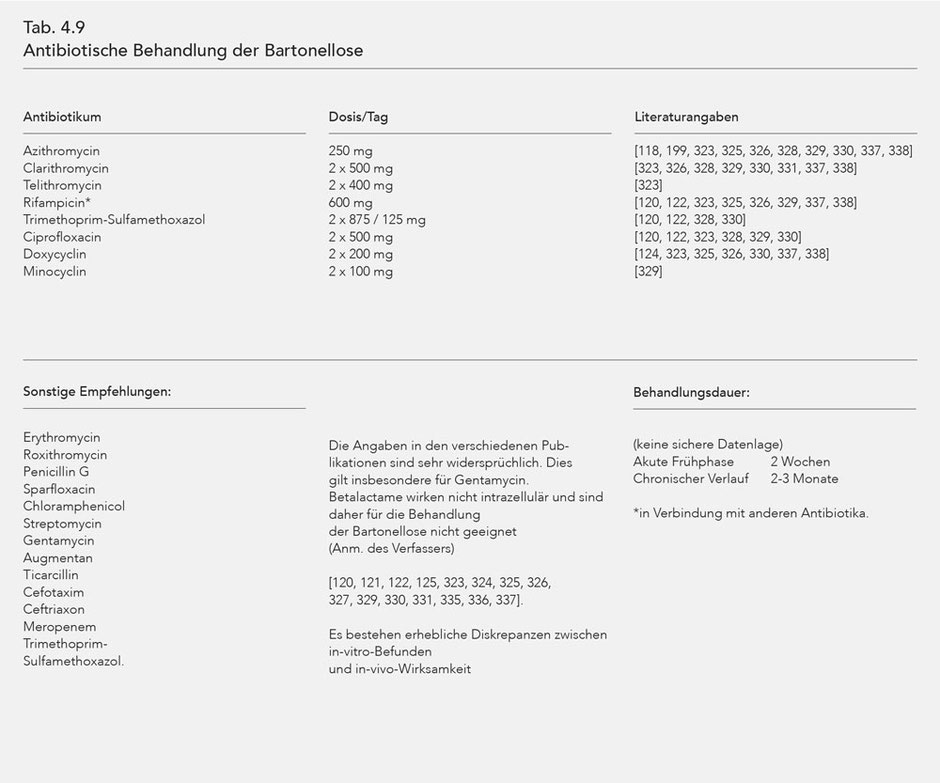
The antibiotic treatment is given in Table 4.9. The following antibiotics are recommended: azithormycin [118, 199], rifampicin, ciprofloxacin, trimethoprim + sulfamethoxazole, gentamycin [120, 122], gentamycin i.v. [121], doxycycline + gentamycin [124, 125].
The treatment is based, in part, on expert recommendations [126]. Other references are listed in Table 4.9.
The discrepancy between the in vitro findings and the in vivo results is set out in various publications [324, 329, 331, 333, 336].
Since Bartonella henselae is primarily localized intracellularly in vivo, only antibiotics that act intracellularly are used; they are listed in Table 4.9.
Because instances of non-response and recidivation are not seldom in cases of chronic bartonellosis, long-term antibiotic treatment is recommended [322, 323, 324, 333, 336].
The efficacy of aminoglycosides (particularly gentamycin) is extremely controversially assessed. Publications with a positive assessment [324, 328, 330, 336, 337] are in opposition to other contributions which assess aminoglycosides as ineffective or insufficiently effective [325, 326, 329].
Chlamydophila pneumoniae
Chlamydophila pneumoniae is important in the differential diagnosis of Lyme disease because of the following disease manifestations.
- Disorders of the nervous system
- Reactive arthritis
- Myocarditis
Chlamydias have special microbiological characteristics: The size of the pathogen is very small compared to other bacterial strains; reproduction occurs only within the host cell; the pathogen is dependent on the host cell’s ATP because it is not able to produce its own.
The pathogen exhibits two phenotypes:
- Elementary bodies
- Reticulate bodies
The elementary bodies can exist extracellularly and are the infectious form. Reproduction of the elementary bodies is only possible in the host cells. After penetration, the elementary bodies are phagocytized by the host cell; intracellularly the elementary body changes into the reticulate body and as such can again divide. The elementary bodies are thus infectious, and the reticulate bodies are reproductive. Some reticulate bodies change back to elementary bodies, which are released subsequent to lysis of the host cell. The thus-produced elementary bodies then infest further host cells. Consequently, the precondition for an effective antibiosis is that the antibiotic is both intracellularly and extracellularly effective. This is the case for tetracyclines and macrolides.
The infection data for Chlamydophila pneumoniae are summarized in Table 4.10.
The primary disease manifestation of Chlamydophila pneumoniae is pneumonia. The incidence is 1% and predominantly affects people older than 65 years of age [132, 133]. The pneumonia is frequently accompanied by infections of the upper respiratory tract (pharyngitis, laryngitis, sinusitis). Slight disease expression initially, extrapulmonary manifestations (see Tab. 4.11) and a normal leukocyte count indicate an atypical pneumonia and thus also pneumonia caused by Chlamydophila pneumoniae.
In addition to pneumonia, Chlamydophila pneumoniae causes extrapulmonary manifestations [134] (cf. Table 4.11), which are significant with regard to the differential diagnosis of Lyme disease (LD) or Lyme neuroborreliosis (LNB).
The chronic course of Chlamydophila pneumoniae infections is documented by studies [234 - 240]. A relationship to CP was also described for Alzheimer’s disease [241]. This is a finding that is also of importance with regard to chronic LNB, for which the same associations have been demonstrated [295 - 298].


The extrapulmonary manifestations frequently extend across a long period of time, i.e. across months and years. This also applies to the so-called reactive arthritis, whose differentiation from the arthritides in Lyme disease is sometimes difficult. Attention should also be paid to the Guillain-Barré syndrome which can last for months and presents in the same manner as in the Lyme disease. The association with a myocarditis is also similar, whereas meningoencephalitis occurs in the acute phase, i.e. practically simultaneously with pneumonia.
The laboratory diagnostics for Chlamydophila pneumoniae includes serology, the lymphocyte transformation test (LTT), and the detection of the pathogenic organism using PCR (Tab. 4.12):
The serology results have severe inherent limitations. There is a considerable discrepancy between the serological findings, on the one hand, and pathogen detection using PCR, on the other hand [135, 136].
A single test for IgG has only a very low sensitivity [137], whereas the sensitivity is quite good in cases of a definite increase in IgG between the acute phase and the further course of the disease.
The diagnostic value of LTT for Chlamydophila pneumoniae has not yet been validated in the literature.
The chronic disease course obviously represents a chronic persistent infection. Chlamydophila pneumoniae could be detected both in the synovial fluid and in the liquor using PCR [234, 235, 236, 239, 240].
The antibiotic treatment of Chlamydophila pneumoniae is given in Table 4.13. The drug of choice is doxycycline; macrolides also exhibit good efficacy, particularly azithormycin; quinolones have a low efficacy [138]. However, gemifloxacin has proven to be very effective [242].


Chlamydia trachomatis
The microbiological anomaly of Chlamydia was presented in the chapter “Chlamyophila pneumoniae”. With regard to the antibiotic treatment, the fact that Chlamydia are present in their infectious form both intracellularly and extracellularly is decisive.
Chlamydia trachomatis is sexually transmitted and causes a urogenital infection. The differential diagnostic reference to Lyme disease results primarily from the arthritides, which are caused by a chronic persistent infection in both diseases. In Chlamydia trachomatis, the arthritis is assigned to the so-called reactive arthritis even though the detection of the pathogen occurred in the synovial fluid in studies [243, 244].
The arthritis occurs in 1% of the urethritis induced by Chlamydia trachomatis. Reiter’s triad (arthritis, uveitis, urethritis) occurs in 0.3% of those affected.
The disease can be easily detected by means of laboratory diagnostics (cf. Tab. 4.14) in cases of existing urogenital infection. In this context, NAATs (nucleic acid amplification techniques) in urethral smear or in urine are available; this examination is also reliable for asymptomatic patients [140-142]. PCR also has a high sensitivity and specificity [143].
The diagnostic value of serology and LTT has not been validated. Moreover, whether a chronic infection with Chlamydia trachomatis (as is the case with LD) can be accompanied by seronegativity has not been clarified. Seropositivity can indeed provide evidence of a previous infection, but principally does not allow any statement with regard to a disorder as a consequence of a persistent infection with Chlamydia trachomatis. Theoretically, a persistent or reproducible pathological lymphocyte transformation test indicates a prolonged infection, but scientific data for the diagnostic value are not yet available.
The significant data on the mode of transmission, symptomatology and treatment are summarized in Table 4.15.


Yersinia enterocolitica infection (yersiniosis)
In the differential diagnosis of Lyme disease and with regard to coinfection, the importance of yersiniosis is primarily based on the disease manifestation of a so- called reactive arthritis. As in Chlamydia infections and probably also in the bartonellosis, the arthritis is very probably the consequence of a chronic persistent infection [163, 164]. Since the so-called reactive arthritis in yersiniosis occasionally also occurs in the scope of Reiter's triad, i.e. in connection with urethritis and uveitis, the autoimmune processes are to be discussed with the pathophysiology. The thyroiditis which frequently occurs in yersiniosis and which is very probably expression of an autoimmune phenomenon as in LD is indicative of such an association.
The anamnestic research subsequent to the early phase of yersiniosis makes an important contribution to the recognition of chronic yersiniosis. The early phase of yersiniosis is essentially characterized by two disease manifestations:
- Gradually beginning gastroenteritis
- Pharyngitis
The infection data and symptomatology of yersiniosis are compiled in Table 4.16.

Yersinia enterocolitica was already recognized as pathogen as early as the beginning of the 20th century. However, the true significance of the pathogen, in particular, under epidemiological aspects was first described in 1995 [146].
Acute illness due to Y. enterocolitica is subject to registration (according to German law).
The pathogen penetrates into the intestinal wall and the mesenteric lymph nodes. Surface proteins and plasmid-bound virulence factors suppress the immune system of the host organism [147-150].
The disease results primarily in gastroenteritis, pseudoappendicitis, und mesenteric lymphadenitis.
In contrast to other bacterial gastroenteritides, the Yersinia enterocolitic gastroenteritis develops gradually and often becomes stressful or perceivable only after a week has passed [15 - 153]. Frequently, the infection is associated with a pharyngitis because the pathogens remain in the lymphatic tissue of the tonsils and the pharyngeal wall, where they can also be detected by means of a smear test. The concurrent occurrence of gastroenteritis with pharyngitis is typical for a yersiniosis [154].
The mean disease duration is approximately two to three weeks, but distinctly longer diseases courses have been described. The acute illness can be associated with numerous gastrointestinal complications, primarily as a consequence of a severe bacterial inflammation of the intestinal wall [155-157]. In addition, the disease can also affect many non-gastrointestinal organs [155, 156, 158 - 161].
The patients frequently remain excretory for months, even when the gastroenteritis has long since abated [152].
The yersiniosis can result in so-called reactive arthritis and is thus an important infectious disease in the differential diagnosis of Lyme disease. Since the disease can also sporadically occur [152] and frequently remains unrecognized, the anamnestic research for typical yersiniosis disease manifestations and data, particularly symptoms of the early phase is of considerable importance, particularly in the early phase.
Differentiation between LD and yersiniosis is made even more difficult by the fact that both infections can cause a multisystem disease. For information on the individual disease manifestations see Table 4.16.
The study by Saebo und Lassen [246], which determined many disease manifestations in a retrospective study of 458 patients, is of particular importance for the depiction of chronic yersiniosis: chronic persistent arthralgias, ankylosing spondylitis, rheumatoid arthritis, iridocyclitis, chronic abdominal pains, chronic diarrhea, ulcerative colitis, nervous disorders, nephritis, thyroid disorders, insulin- dependent Diabetes mellitus, chronic hepatitis, (multisystem diseases) and a substantial reduction of the overall life expectancy. Many of the different relationships were depicted in further publications by these authors [252 - 256, 257].
Studies which immediately suggest a possible relationship between Yersinia and inflammatory intestinal disorders [257] close the pathophysiological circle between Yersinia, inflammatory intestinal disorders and enteropathic arthritides.
Despite this, it should be noted that the relationship between Yersinia infection and the above-mentioned numerous disease manifestations (except for arthritis) have been inadequately analyzed. This may be due to the fact that yersiniosis’ significance as a disease has only been recently recognized.
The so-called reactive arthritis primarily affects the hip, knee and upper ankle joints as well as the sacroiliac joints; occasionally there are additionally chronic pains in the lumbosacral region [165]. This arthritis can last for months, and exhibit recurrent and symptom-free intervals in the disease course. — The so-called reactive arthritis in yersiniosis can occur alone, but occasionally also in connection with conjunctivitis and urethritis (previously termed Reiter's syndrome [162]).
In the differential diagnosis of Lyme disease, it is of particular interest that the pathogen (Y. enterocolitica) could be detected in articular effusions in studies of the so-called reactive arthritis [163, 164].
Sometimes these arthritides last for many years. In addition, there is a relationship between yersiniosis and thyroiditis. All these facts (chronic arthritides, multisystem disorders, disease course lasting for years, correlation with regard to thyroiditis) can be observed in the same manner in Lyme disease. Thus, the differential diagnosis is sometimes extremely difficult.
The laboratory diagnostics of Yersinia enterocolitica infection is presented in Table 4.17.
As is the case in Lyme disease, there is often seropositivity in non-diseased people Information on a possible seronegativity in chronic yersiniosis is not available.
In the disease course the serological findings can correlate with the disease expression [165].

It is not at all seldom that a highly significant pathological Yersinia LTT is found in patients whose complaint symptomatology is primarily consistent with chronic Lyme disease. In correspondence to chronic Lyme disease, the positive yersinia LTT could be an indication of a chronic persistent infection especially in cases involving reproducibility.
The focuses which are to be considered in the differential diagnosis of chronic Lyme disease or chronic yersiniosis, respectively, are depicted in Table 4.16.
Pathogen detection is particularly possible in articular effusion as well as in the lymphatic tissue of the intestine as well as in early stages also by means of a throat swab. Data on the sensitivity of pathogen detection using PCR or culture methods does not exist in the relevant literature.
In Yersinia-PCR-positive patients, the serology was positive in 70% of the cases; the LTT in 50% [248].
In the initial detection of Yersinia enterocolitica using culture methods, IgA and IgG bands were found in immunoblot assay in patents experiencing a chronic course. The continuous detection of IgA antibodies was obviously an expression of a persistent infection; in this context the pathogens were detected in the intestinal mucosa and in lymphatic tissue. Hence, this was a definitely chronic, persistent Yersinia enterocolitica infection [258]. The antibiotic treatment of the Yersinia enterocolitica infection is presented in Table 4.18.

Yersiniosis frequently abates within a few weeks so that an antibiotic treatment is not generally recommended. This also applies with regard to the excretors. Only in cases of severe disease courses, in particular with sepsis, are antibiotics used.
Y. enterocolitica produces beta-lactamases with the consequence that penicillin, ampicillin and the cephalosporins of the first generation are ineffective [201, 205]. There is also frequently a resistance to macrolides.
It is also disputed whether early antibiotic treatment (i.e. for gastroenteritis) prevents reactive arthritis [203].
The differential diagnosis of chronic yersiniosis or chronic Lyme disease, respectively, is thus as a consequence of the many instances of overlap in the symptomology extremely difficult. In cases in which both diseases are present in their chronic form, a differentiation is often not possible at all.
Mycoplasma pneumoniae infection
The differential diagnostic differentiation between LD und Mycoplasma pneumoniae infection or the recognition of the coinfection by Mycoplasma pneumoniae is problematical because both diseases
exhibit many identical disease manifestations; this applies to the extrapulmonary manifestation in Mycoplasma pneumoniae infections: disorders of the CNS, of the musculoskeletal system, of the
heart, of the kidney and of the eye.
The infection data and the symptomatology are given in Table 4.19. In the foreground is the atypical pneumonia, frequently linked to symptoms in the region of the upper respiratory tract. There is no data on the frequency of extrapulmonary manifestations in the literature.
Mycoplasma pneumoniae is considered to be the most important pathogen of atypical pneumonia. However, pneumonia only occurs in approximately 3% - 10% of the cases in Mycoplasma pneumoniae infections [204]. In most cases, the infection results in a banal bronchitis [204], pharyngitis, rhinitis, ear aches, and sinusitis [205].
All the extrapulmonary disease manifestations listed in Table 4.17 are seldom [206 - 214]. In arthritis, Mycoplasma pneumoniae was detected in the synovial fluid by
means of PCR [211]; this is an indication of a direct relationship to the infection.
Pathogen detection in articular effusion and the many extrapulmonary disease manifestations document the chronic disease course in cases of Mycoplasma pneumoniae. However, precise data on the chronic disease course are not available in the literature In particular, whether a chronic infection, especially with extrapulmonary disease manifestation, can persist with seronegativity is unclear. Seropositivity documents the infection, but it principally cannot serve as a diagnosis basis for a chronic persistent Mycoplasma pneumoniae infection.
The literature on the relationship between Mycoplasma pneumoniae and neurological disease manifestations is comparatively extensive. The publications primarily refer to neurological complications in pneumonia, i.e. the early phase of the Mycoplasma pneumoniae infection.
The neurological manifestations involve both the early phase, i.e. the point in time of existing pneumonia due to Mycoplasma pneumoniae, and later disease stages. Changes in the region of
the brain stem [259, 267], myelitis [260, 263, 265, 269, 271, 274, 277, 279, 281, 284, 286], Guillain-Barré syndrome [261, 262, 268, 272, 282, 283], encephalitis [270, 273, 275, 276, 278, 280,
281, 285, 286], meningitis [270], polyradiculopathy [263], peripheral

facial paresis [264, 266], optical neuritis and hemorrhagic leukoencephalitis [268], peripheral polyneuropathy [270], disorders of the brain nerves [282], radiculitis [282] have been described.
The frequency of neurological symptoms in connection with Mycoplasma pneumoniae varies between 1‰ [287], 1% [288], and 5% [289]. The pathogen has been repeatedly detected by means of culture methods or PCR [270, 274, 283].
Pathogen detection in serum and liquor is considered to be proof that the neurological manifestations are mediated infectiously and not immunologically [283]. However, the connection between Mp and neurological manifestations is not undisputed [290, 287].
Other extrapulmonary manifestations mentioned in the literature involve hepatitis, hemolytic anemia, Schönlein-Henoch purpura, disorders of the muscular-skeletal system, of the skin and other organs [265], macula edema [270], bilateral uveitis [291], nephritis [292], arthritis, hepatitis, pericarditis [292].
The laboratory diagnostics for Mycoplasma pneumoniae is presented in Table 4.20. The serology only becomes positive after several weeks, as is the case for most infectious diseases. It is therefore significant for the chronic disease course. Seropositivity substantiates the infection, but not the disease. Whether a chronic infection can also exist in cases of seronegativity has not yet been scientifically clarified.
The LTT for Mycoplasma pneumoniae has not been validated by studies.
Pathogen detection, e.g. in articular effusion is possible, but it is difficult, has a low sensitivity and is therefore not part of routine diagnostics.
The antibiotic treatment of Mycoplasma pneumoniae is presented in Table 4.21. The drugs of choice are azithormycin [293] und levofloxacin [294].


Secondary coinfections of Lyme disease
In the following, additional infections will be presented, which are mentioned in international publications as coinfections of Lyme disease—in particular HGA (human granulocytic anaplasmosis) and babesiosis. These two coinfections are of importance in the USA, but not in Europe.
Because of the similarity of their symptoms to those of LD, the following diseases are included for completeness’ sake: tularemia, Q fever, parvovirus B19 infection and Campylobacter jejuni infection.
Human granulocytic anaplasmosis (HGA)
Human granulocytic anaplasmosis (Synonym: Human granulocytic ehrlichiosis (HGE)) is transmitted by ticks. Its reservoirs are red deer and the white-footed mouse The HGA pathogen can be simultaneously transmitted with Borrelia burgdorferi with the consequence of a double infection. HGA exhibits many symptoms which also occur in the same form in LD. Indications of HGA are pathological laboratory findings in the form of leukopenia, thrombocytopenia, and elevated transaminases.
The pathogen is localized intracellularly. The transmission to mice has been proven [193].
In connection with ehrlichiosis or anaplasmosis, respectively, two pathogens are to be noted:
- Ehrlichia chaffeensis [1]
- Anaplasma phagocytophilum [2]
E. chaffeensis infects monocytes; A. Phagocytophila, granulocytes.
E. chaffeensis is the pathogen of human monocytic ehrlichiosis (HME), a very rare infectious disease, which occurs primarily in the USA and some regions of South America, but practically nowhere else on earth.
E. phagocytophila is the pathogen of human granulocytic anaplasmosis, another extremely rare disease in the USA with an annual incidence of approximately 10 / 1 million in habitants [3].
The infection data and symptomatology are compiled in Table 4.22.
The pathological importance of human monocytic ehrlichiosis (HME) and human granulocytic anaplasmosis (HGA) was discovered in 1986 and 1994, respectively [35, 36]. The two infectious diseases resemble each other clinically and with regard to laboratory findings.
The pathogens develop in monocytes (HME) or in granulocytic leukocytes (HGA). Thus, their localization is exclusively intracellular.
The pathogens are transmitted by ticks, in the United States primarily via Ixodes scapularis, in Europe, via I. ricinus.
Important reservoirs: deer (HME); wood mice (HGA) In addition, other modes of transmission are being discussed:

mother-child transmission, blood transfusions, direct contact with infected animals, transmission form person to person [37, 38, 41, 42, 43, 44].
Scientific reports on illnesses due to HGA in Europe are rarities [4]. However, studies in Northern Italy showed that 24% of the ticks (I. ricinus) were infected by E. chaffeensis or A. phagocytophilum. Similar figures have been substantiated in the Netherlands and in Poland, whereas in Germany they are at approximately 2% [5 - 10]. The figures for the East Coast of the United States were higher; approximately of the order of 30% - 40% [11, 12].
In patients with Lyme disease the seroprevalence for A. phagocytophilum in Europe is approximately 10% [13 - 15]. Similar figures were also obtained in the USA [16].
Since seroprevalence merely expresses something about the frequency of the infection, but nothing about the disease (HGA), no reliable statements about the frequency of the disease (prevalence of HGA) can be made. According to the laws of probability, a concurrent HGA infection in patients with Lyme disease might amount to a few percent at most.
There is no literature on chronic HGA courses. However, subacute and chronic courses are discussed [17, 19].
The incubation period, i.e. the time between the tick bite and emergence of the acute illness, is approximately one week on average [18].
The laboratory diagnostics for HGA is presented in Table 4.23. As mentioned above, HGA is characterized by leukopenia, thrombocytopenia, and elevated transaminases. Such changes occur frequently and are—particularly in cases involving a febrile clinical picture with the above-mentioned symptoms (Tab. 4.22)—an indication for HGA. Verification of the infection is performed using serology, other methods of detection, particularly direct pathogen detection, are not often successful and therefore are not part of the routine diagnostic methods.

The determination of HGA (also as coinfection) merely on the basis of serological findings is dubious since seropositivity does not verify the presence of the disease (HGA), but only the prior infection. Also in the case of HGA, the diagnosis is based primarily on the totality of anamnesis, physical examination findings, medicinal- technical findings, and differential diagnosis.
Doxycycline is recommended as therapy, also for children. Precise literature with regard to an adequate treatment is not available.
In summary, it can be stated that HGA as individual disease and as so-called coinfection in cases of Lyme disease is not of major importance in Europe; however, the literature in this problem complex is currently completely inadequate.
Babesiosis
Pathogens: Babesia microti, Babesia divergens [57, 58]
Vector: ticks (I. ricinus (Europe), I. scapularis (USA)) [57, 58]
Other modes of transmission: blood transfusions [59], perinatal [60, 61]
Reservoir: cattle (other vertebrates)
Laboratory diagnostics:
- Detection in blood smear (difficult, repetition frequently required)
- PCR (higher sensitivity than blood smear [62])
- Serology [63, 64]
- Poor correlation between serologic titer and symptomatology [64]
Babesia are protozoa and result in lysis subsequent to invasion of erythrocytes.
Two species of Babesia are pathogenetically significant.
- Babesia microti
- Babesia divergens
B. microti is the predominant pathogen in the USA; B. divergens, in Europe [cf. 196]. The transmission of the pathogen occurs primarily via ticks. B. microti has been found as a coinfection in LD [197, 198, 199].
Since 1956 a total of only 30 cases has been reported in Europe. The majority of these patients were splenectomized.
The prevalence of B. microti and B. divergens in ticks is 10% - 20% in Europe [21 - 23], in the USA, sometimes higher [24].
The seroprevalence with regard to B. microti and B. divergens is 0% in patients with Lyme disease [25, 26] and thus is in stark contrast with the frequency of the pathogen in ticks.
The situation in the USA is different: there the seroprevalence is approximately 10% - 20% [27 - 30]. In the USA instances of the disease are reported correspondingly more frequently, in some cases with severe disease courses [31 - 34]. This difference can obviously be only explained by the fact that B. microti, the predominant pathogen in the USA, has a very much higher virulence than B. divergens.
Thus, Babesiosis dos not play a major role in Europe unless the patient contracted the disease in a foreign country, e.g. in the USA.
The clinical picture presents as a febrile, influenza-like medical condition with chills and fever, arthralgias, myalgias, and gastrointestinal symptoms. Severe disease courses only occur in non-immunocompetent patients.
On base of own experiences chronic Babesiosis in the European area cannot be excluded definitely. The disease is characterized by the following symptoms:
- marked fatigue
- chronic sickness
- slight fever
- air hunger
- dyspnea
- headache
- myalgia
- arthralgia
- loss of appetite
- neckstiffness
- sore throat
- unproductive cough
- weight loss
- nausea
- vomiting
- diarrhea
- haemolytic anemia
- thrombocytopenia
- elevated transaminases
The treatment is carried out with atovaquone, azithormycin, clindamycin, if necessary in combination with quinine.
However, by means of the assessment of all data, it can be determined that babesiosis—due to the dominant European pathogen, B. divergens—does not represent a major health hazard and thus is of little consequence as a coinfection in cases of Lyme disease.
Literaturübersicht Babesiose
Krause PJ et al., 1998 [359].
46 Patienten, infiziert mit Babesia, Connecticut, Patienten mit akuter Babesiose, Untersuchung mittels Blutausstrich und PCR. Unbehandelt kann die Babesiose über
Monate und sogar Jahre persisieren. Clindamycin und Chinin reduzieren die Parasitämie, jedoch kann die Infektion persistieren. Bessere Behandlungsmethoden werden benötigt.
Hatcher JC et al., 2001 [360].
34 Patienten, hospitalisiert wegen schwerer Babesieninfektion. – Symptomatik: Allgemeines Krankheitsgefühl, Arthralgien, Myalgien, Dyspnoe, Thrombozytopenie, abnorme
Leberfunktion. Behandlung erfolgte mit Kombinationstherapie, verwendet wurden Clindamycin, Chinin, Atovaquon oder Azithromycin. Trotz Behandlung persistierte die Parasitämie 8,5 Tage (3-21
Tage).
Hunfeld KP et al., 2002 [361].
467 Seren von Patienten mit Lyme-Borreliose (Erythema migrans), Bb-seropositiven beschwerdefreien Patienten, Personen mit anamnestisch angegebenem Zeckenstich und
gesunde Kontrollpersonen. Personen, die Kontakt mit Zecken hatten, wiesen eine signifikant höhere Seroprävalenz für Babesia auf. Alle Kollektive zusammengenommen zeigten eine Seroprävalenz für B.
microtii von 5,4%, für B. divergens 3,6%. Die Ergebnisse zeigen, dass Infektion mit Babesia eine Coinfektion der Lyme-Borreliose sein kann und dass Babesien-Infektionen in der BRD häufiger
vorkommen als bisher eingeschätzt.
Oleson CV et al., 2003 [362].
74-jähriger Patient mit Querschnittsmyeliltis infolge Lyme-Borreliose und Babesiose. Babesien nachgewiesen im Blutausstrich und mittels PCR. LB-Serologie positiv.
Zunächst Tetraplegie, nach 2 Monaten (nurmehr) Paraplegie.
Hildebrandt A et al., 2007 [363].
42-jährige Patientin mit akuter myeloischer Leukämie. Nachweis von Babesia microti mittels PCR. Erster in Europa bestätigter Fall einer autochtonen Babesiose.
Hunfeld KP et al., 2008 [364].
Übertragung von Babesia durch Zecken. Symptomatik: Erythrozyten-Lyse, Anämie, Hyperbilirubinämie, Hämoglobinurie und mögliches Organversagen.
Ohmori S et al., 2011 [365].
PCR Methode zum Nachweis von Babesia, auch Unterscheidung von verschiedenen Subspezies möglich.
Krause PJ, 2003 [366].
Symptomatik: Anämie, Thrombozytopenie. Diagnose durch Blutausstrich. Nur 1% der Erythrozyten sind befallen. PCR hat vergleichbare Sensitivität und Spezifität.
Ergänzend ist serologische Untersuchung sinnvoll. Behandlung: Clindamycin (600 mg/6 Stunden), Chinin (650 mg/8 Stunden), Azithromycin (ca. 250 mg/Tag), Atovaquon (750 mg/12 Stunden). Azithromycin
und Atovaquon haben geringere Nebenwirkungen. Eine Austauschtransfusion kann lebensrettend sein.
Häselbarth K et al., 2007 [367].
Erster Fall einer Babesiose bei Menschen in BRD. 63-jähriger splenektomierter Patient mit rezidivierendem nodulären Hodgkin-Lymphom. Nachweis von Babesia durch
Blutausstrich und PCR. Behandlung Chinin, Clindamycin nicht erfolgreich. Bei Rückfall Nachbehandlung als Langzeitbehandlung mit Atovaquon.
Wormser GP et al., 2010 [368].
3 Immun-defiziente Patienten mit Babesiose. Behandlung mit Azithromycin, Atovaquon. Während der Behandlung Entwicklung einer Resistenz.
Hunfeld KP et al., 1998 [369].
Serologische Untersuchung bezüglich HGE- und Babesia microti Serologie. Bei LB Serologie Babesia microti positiv in Stadium I und II bei 13-18%. HGE Serologie bei
3-7%. In der Spätphase Serologie für HGE und Babesia microti negativ.
Rickettsioses
To begin with, attention should be directed to the fact that the bartonellosis pathogen belongs to the Rickettsia family.
In the USA the most important rickettsiosis is Rocky Mountain Spotted Fever (RMSF), a potentially fatal, but normally curable disease. RMSF is the most frequent rickettsiosis in the USA. The clinical picture is primarily characterized by high fever, pronounced malaise, abdominal complaints, and a generalized exanthema. Occasionally, the disease is also linked with CNS manifestations (focal neurological deficits, cerebral seizures):
Worldwide, there are many different rickettsioses, which are caused by different Rickettsia subspecies. The transmission generally occurs via ticks, but also via mites, fleas, and lice. Generalized exanthema—the so-called localized eschar (black wound)—as well as fever, headaches and severe muscle pains are typical symptoms of the disease.
The most important rickettsiosis in Europe is the Mediterranean spotted fever; pathogen: R. conorii. The disease primarily affects Southern Europe.
Treatment is performed with doxycycline.
Chronic courses have not been described in the literature. Differential diagnostic problems in comparison to Lyme disease in the early stage should not result because of the endemic circumstances and the presence of the exanthema.
For further information refer to the relevant medical literature.
Tularemia
Tularemia is caused by the pathogen Francisella tularensis. Transmission occurs via mosquitos. The disease reservoir comprises many vertebrates.
Main disease manifestations: fever, headaches, malaise, swollen lymph nodes, pharyngitis, eschar (black wound), emesis, pneumonia, erythematosus papular- ulterative lesion at the location of the bite with black spot (central eschar, “tache noire”).
Treatment: tetracyclines, ciprofloxacin.
Betalactamases are ineffectual.
Relapses can occur; persistent chronic courses have not been described in the relevant literature. In the differential diagnosis problems can occasionally occur in the early stage in cases lacking erythema migrans in comparison with Lyme disease.
For further information refer to the relevant medical literature.
Q fever
Q fever is caused by the pathogen Coxiella burnetii. In contrast to rickettsioses (Mediterranean fever) and tularaemia, Q fever can exhibit a chronic course. There are differential diagnostic problems in distinguishing it from late Lyme disease (Stage III) in the symptomatology.
Q fever generally occurs endemically (transmission from human to human). The transmission primarily occurs via contact with infected livestock namely by inhalation or oral transmission of the pathogen. Admittedly, C. burnetii can also be found in other reservoirs, e.g. in ticks, but two items are decisive for the diagnosis:
Endemic occurrence (contact with people suffering from the disease).
Contact with (diseased) livestock and their products (milk products)
The decisive diagnostic information with regard to a possible Q fever is thus the person’s occupational activity or the contact with agriculture and livestock. In sporadic cases, frequent consumption of raw milk or contact with diseased cattle (abort) can be an indication of an infection hazard.
Significant disease manifestations:
- Transitory influenza-like clinical picture
- Pneumonia
- Hepatitis
- Other manifestations:
-
- Erythema
- Pericarditis / Myocarditis
- Meningitis / Encephalitis [318-320]
- Myelitis [316, 317]
- Chronic fatigue [340-345]
The chronic course of Q fever can persist for months or years. In this context, chronic endocarditis is the predominant disease manifestation. A chronic course occurs in approximately 1 to 5% of those patients who were afflicted with an acute Q fever. In addition to the endocarditis, infection of aneurysms and vascular prostheses is a frequent occurrence.
The entire symptomatology of chronic Q fever is presented in Table 4.24 [315, 345, 347-350].
Pregnancy is a risk for the development of chronic Q fever. Untreated chronic Q fever results in a substantial morbidity and in mortality up to 60% [350].
Chronic Q fever requires a long-term antibiotic treatment, initially with doxycycline + hydroxychloroquine [350]. The antibiotic treatment must be performed for a period of at least 18 months [350].
The diagnosis is supported by serological tests. If the IFA IgG is 1:800 or higher, chronic Q fever cannot be ruled out. However, the laboratory diagnostics are not yet adequately standardized. The detection of the pathogen using PCR in tissue (e.g. heart valves) has a sensitivity of 100%, in blood of 47%; the sensitivity in buffy coat is somewhat higher. The examination of heart valves using echocardiography and, if necessary, FTG-PET, CT, MRT is also of diagnostic importance.
In 25% of the chronic Q fever cases heart valve changes can be detected echocardiographically.
Wegdam-Blans et al [350] have developed diagnostic guidelines for chronic Q fever (Tab. 4.25):
Frequent follow-up examinations are required during the disease course.
On suspicion of chronic Q fever:
- Echocardiography
- PET CT scan
Antibiotic treatment of chronic Q fever:
- Doxycycline
- Hydroxychloroquine
- Macrolides
(azithromycin, clarithromycin, telithromycin) -
- Fluoroquinolone
- Trimethoprim and sulfamethoxazole
[345, 347-348]


In cases of chronic Q fever, treatment with doxycycline + hydroxychloroquine as a long-term treatment, i.e. for 18 months, is recommended [350].
Zur Vervollständigung wird im Folgenden die Darstellung der Problematik durch die CDC und die Q Fever Working Group, 2013 eingefügt.
Diagnosis and Management of Q Fever –
United States, 2013:
Recommendations from CDC and the Q Fever Working Group.
Einleitung:
Coxiella burnetii, intrazellulär, vor allem in mononukleären Phagozyten aber auch in anderen Körperzellen. Übertragung durch Inhalation von verseuchter Luft (Exkrete
infizierter Tiere). Andere Übertragungswege: Zecken, unpasteurisierte Milch, Milchprodukte, sexuelle Übertragung. Diagnose durch serologische Untersuchung. Behandlung: Doxycyclin.
Epidemiologische Faktoren:
- Berufliche Tätigkeit
- Kontakt mit Vieh
- Aufenthalt in endemischen Gebieten
- Sexualkontakt mit Q-Fieber-Patienten
- Familienmitglied mit Q-Fieber
- Anamnese eines akuten Q-Fiebers
- Patienten mit Herzklappenfehlern und vaskulären Prothesen
Akutes Q-Fieber:
Erwachsene:
- Inkubation 2 bis 3 Wochen
- Pneumonie
- Hepatitis
- Fieber
- Fatigue
- Schüttelfrost
- Myalgien
- Heftige Kopfschmerzen
- Photophobie
- Myalgien
- Arthralgien
- Makulo-papuläres oder purpurfarbenes Exanthem
- Pericarditis
- Myocarditis
- Aseptische Meningitis
- Encephalitis
- Cholezystitis
Kinder:
- Gastrointestinale Beschwerden (50-80%)
- Hämolytisches urämisches Syndrom
- Lymphadenitis
- Cholezystitis
- Rhabdomyolyse
- Sonstige Symptome wie bei Erwachsenen
Schwangere:
- Frühgeburt
- Intrauterine Fehlentwicklung (bei Infektion im ersten Trimester)
- (Fehlbildungen durch Q-Fieber sind in der Literatur nicht beschrieben)
- Infektion bei Schwangerschaft: Doxycyclin
Laborbefunde:
- GPT erhöht (85% der Fälle)
- Thrombozytopenie
- Nachfolgend Thrombozytose
- BSG erhöht
- CRP erhöht
- Hyponatriämie
- Hämaturie
- CK erhöht
Chronisches Q-Fieber:
5% der Fälle mit akutem Q-Fieber entwickeln chronisches Q-Fieber. Beginn Monate, Jahre oder Jahrzehnte nach akuter Infektion oder nach asymptomatischem akutem Q-Fieber.
- Endocarditis
- Hepatitis
- Chronische vaskuläre Infektion
- Osteomyelitis
- Osteoarthritis
- Chronische pulmonale Infektion
Endocarditis führende Problematik.
Sonstige Symptome:
- Fatigue
- Fieber
- Abdominelle Schmerzen
- Thorakale Schmerzen
- Gewichtsverlust
- Nachtschweiße
- Hepatomegalie
- Arterielle Embolie
- Lungenembolie
- Tiefe Venenthrombose
- Vegetationen auf Herzklappen
- (Nachweis durch TEE)
Anmerkung:
Die Fälle mit Lungenembolie traten bei Patienten mit Endocarditis auf, die Fälle mit Thrombose offensichtlich bei schwer erkrankten Patienten. Thrombose und
Lungenembolie kommen bei Q-Fieber selten vor.
Post-Q-Fieber-Fatigue-Syndrom:
Bei 20% der Patienten nach akutem Q-Fieber (Literatur spärlich).
Symptome:
- Übelkeit
- Kopfschmerzen
- Nachtschweiße
- Myalgien
- Faszikulationen
- Lymphadenome
- Arthraglien
- Schlafstörungen
- Alkoholintoleranz
- Photophobie
- Nervosität
- Reizbarkeit
- Depression
- Kognitive Störungen
Dauer über ein Jahr, oft mehrere Jahre oder lebenslang.
Pathogenese unklar.
Leitlinien (Empfehlungen) bezüglich Behandlung liegen nicht vor.
Laboruntersuchungen
Akutes Q-Fieber:
Phase II AK treten zunächst auf und sind höher als Phase I AK.
Diagnose gesichert:
Vierfacher Anstieg des Phase II IgG im IFA
Verglichen werden die Werte in der Akutphase mit denen 3 bis 6 Wochen später
In der ersten Woche AK oft nicht vorhanden, so dass Entscheidung für Behandlung schwierig ist.
Serokonversion tritt nach ein bis zwei Wochen auf. 90% der Patienten sind in der dritten Krankheitswoche seropositiv. IgG Phase II über 1:128 indiziert akutes
Q-Fieber.
IgM-AK von begrenztem diagnostischen Wert.
Bei Verdacht auf Q-Fieber sollte umgehend antibiotisch behandelt werden, also Behandlungsbeginn nicht abhängig machen von Laborbefunden. Mittel der Wahl:
Doxycyclin.
Erregernachweis mittels PCR im Blut möglich. PCR positiv bei fast allen Patienten mit akutem Q-Fieber (Erregernachweis mittels PCR möglich, bevor AK
auftreten).
Chronisches Q-Fieber
Duke Kriterien:
(Die Duke Kriterien betreffen die infektiöse Endocarditis einschließlich Q-Fieber. Für das chronische Q-Fieber gelten die folgenden Kriterien):
Diagnostisch gefordert eine der folgenden Konstellationen:
- 2 Hauptkriterien, 0 Nebenkriterien
- 1 Hauptkriterium + 3 Nebenkriterien
- 0 Hauptkriterien + 5 Nebenkriterien
Hauptkriterien:
- Erregernachweis im Blut
- Echokardiographie: Nachweis von Vegetation an Herzklappen, vaskulären Prothesen, neu aufgetretene Insuffizienz prothetischer Klappen
- Phase I IgG AK Titer > 1:800
Nebenkriterien:
- Prädisponierende Herzerkrankung
- Drogenabusus
- Temperatur über 38 Grad Celsius
- Arterielle Embolien
- Lungeninfarkt
- Intracranielle Blutungen
- Konjunktivale Blutungen
- Palmare Mikroembolien
TEE gefordert
Serologie bei chronischem Q-Fieber:
Phase I IgG > 1:1.024 und möglicherweise höher als Phase II Titer
(Nach Duke-Kriterien Phase I IgG > 1:800 gefordert)
Behandlung: Chronisches Q-Fieber:
Doxycyclin 200 mg + Hydroxychloroquin 600 mg
Dauer der Behandlung abhängig vom Krankheitsverlauf.
Kombination mit Hydroxychloroquin erforderlich
(Hydroxychloroquin erhöht den pH-Wert in den Lysosomen. In vitro nachgewiesen, dass Doxycyclin und Hydroxychloroquin bakterizid auf C. burnetii wirken).
Chronisches Q-Fieber: Behandlungsdauer mindestens 18 Monate bei Herzklappenfehlern und zwei Jahre bei Klappenprothesen.
Sonstige Manifestationen eines chronischen Q-Fiebers werden antibiotisch behandelt in Abhängigkeit vom Krankheitsverlauf.
Die Abnahme von Phase I IgG und die Besserung klinischer Symptome sind Hinweis auf eine effektive Behandlung.
Beschwerdefreie Patienten nach antibiotischer Langzeitbehandlung mit persistierenden Phase I IgG Werten von > 1:1024 haben durch Fortsetzung der antibiotischen
Behandlung möglicherweise keine weiteren Vorteile.
Bei Patienten mit Herzklappenfehler und Q-Fieber serologische Kontrolle (nach Behandlung) halbjährlich für mindestens 5 Jahre, ggf. lebenslang.
Die Behandlung des chronischen Q-Fiebers ist schwierig, sie hängt im Wesentlichen von der klinischen Einschätzung ab.
Zusammenfassung Behandlung Q-Fieber:
Bei Verdacht auf akutes Q-Fieber sofortige antibiotische Behandlung, also nicht auf serologischen Befund warten
Chronisches Q-Fieber nur behandeln, wenn serologische Bestätigung vorliegt
Antibiotische Behandlung nur bei symptomatischen Patienten
Doxycyclin Mittel der Wahl
Behandlungsdauer 2 Wochen
Kinder unter 8 Jahren Behandlung mit Trimethoprim-Sulfomethoxazol, alternativ Doxycyclin für nur 5 Tage
Behandlung in der Schwangerschaft: Trimethoprim-Sulfomethoxazol während der gesamten Schwangerschaft
Serologische Verlaufskontrolle nach akutem Q-Fieber, um ein chronisches Q-Fieber rechtzeitig zu erfassen.
Human parvovirus B19 infection
Pathogen: human parvovirus B19
Transmission: respiratory tract (droplet infection)
Transmission during pregnancy
Blood transfusion
Reservoir: human beings
The disease can exhibit a chronic course, i.e. over months and years [171]. Whether parvovirus B19 causes chronic myocarditis and cardiomyopathy is a matter of dispute [172-175]. The chronic course is verified by means of pathogen detection in articular effusion, myocardium, bone marrow, and blood [169-175].
With regard to Lyme disease, the following differential diagnostic disease manifestations are relevant:
- Persistent or recurrent arthropathy
- Myocarditis
- Cardiomyopathy
Fifth disease (erythema infectiosum) is a typical skin manifestation of a parvovirus B19 infection in children, but does not normally occur in adults. Arthralgias can last for months or years.
For further information refer to the relevant medical literature.
Campylobacter jejuni
Campylobacter jejuni (Cj) is a small gram-negative bacterium whose pathological significance was recognized in the 1980s. Worldwide, Cj is among the most frequent pathogens causing acute diarrhea Sources of infection are game animals and domestic animals, animal products and especially poultry [299]. The pathogen can persist in a coccoid form, but also in its normal form, for months in cases of unfavorable living conditions. It penetrates into the epithelial cells of the intestine and causes their destruction, possibly by means of toxins [303, 304].
The main manifestation of the Campylobacter jejuni infection is gastroenteritis. In the early phase (gastroenteritis), complications can occur in the abdominal region.
Cj has differential diagnostic significance due to complications in the late stage: reactive arthritis, Guillain-Barré syndrome.
Reactive arthritis in Cj infections is seldom; its frequency amounts to a maximum of 2.6% [305 - 308]. In connection with a Cj infection, the Guillain-Barré syndrome has a comparatively unfavorable prognosis [309]. Its incidence amounts to approximately 1‰ [310].
Reactive arthritis occurs approximately one to two weeks after gastroenteritis [304]; Guillain-Barré syndrome, in a period approximately two months after onset of infection [311].
The infection data and symptomatology are compiled in Table 4.24.
Antibiotic treatment reduces the duration of gastroenteritis. Macrolides [312] and quinolones are primarily recommended, but resistances to them can also occur [313]. Resistance to trimethoprim und beta-lactamases exists [314].

Brucellose
Der Erreger Brucella spp. wird bei Kontakt mit tierischen Flüssigkeiten, insbesondere von Haustieren sowie durch Verzehr von infizierten Milchprodukten übertragen. Übertragung von Mensch zu Mensch (B. melitensis, Inhalation) und endemisches Auftreten sind möglich.
Die Brucellose kann zunächst als akutes fieberhaftes Krankheitsbild auftreten und sich bei unzureichender Therapie oder inadäquater Immunabwehr zu einem chronischen
Krankheitsbild entwickeln, mitunter auch in Form von Rezidiven. Die wichtigsten Krankheitsmanifestationen sind in Tab. 4.26a wiedergegeben.
Das akute Krankheitsbild kann plötzlich, jedoch auch schleichend beginnen.
Diagnostisch entscheidend ist die Erkennung im Zusammenhang mit krankem Nutzvieh oder verseuchten Milchprodukten. Brucella ist häufige Ursache für Abort bei
Tieren.
Die Diagnose stützt sich im Wesentlichen auf die Serologie, Erregernachweis ist durch PCR oder Kultur möglich.
Behandlung der Brucellose: Doxycyclin, Streptomycin, Rifampicin, Fluorchinolone (siehe Tab. 4.26a).
Im Übrigen sei auf die Fachliteraur verwiesen.

Reactive arthritis
The term “reactive arthritis” characterizes arthritides which bear a relationship to certain infectious diseases. In former times the term “Reiter syndrome” was used in cases of concurrent
infection of the urethra and the uvea. Arthritis, urethritis and uveitis were termed Reiter’s triad [215, 216]. Infectious diseases which can induce reactive arthritis are given in Table 4.25.
Infektionskrankheiten, die eine Reaktive Arthritis induzieren können, sind in Tabelle 4.27 wiedergegeben.
The term “reactive arthritis” is not a defined disease (nosological entity), but rather a concept for the classification of disease relationships and of the pathophysiology.

The term “reactive arthritis” is problematical because in many infections the pathogens were detected in the synovia and joint fluid in cases of such so-called reactive arthritis. This is true for Chlamydophila pneumoniae [234-236], Chlamydia trachomatis [243, 244] and for Yersinia enterocolitica [163, 164]. — Moreover, the pathogen was also found in the synovia in cases of arthritides in connection with Mycoplasma pneumoniae [211].
Reactive arthritis occurs days to weeks after the onset of infection. It predominantly affects the joints of the lower extremities. In cases involving a disease duration of less than 6 months, the term “acute reactive arthritis” was selected, for disease duration of more than 6 months, the term “chronic reactive arthritis" is used.
In 50% of the cases, joints of the upper extremities are also affected, including the small joints, and the arthritis can be accompanied by cases of tendonitis (enthesitis) [217 - 219, 220].
One of the most important differential diagnoses of reactive arthritis is Lyme arthritis (chronic Lyme disease, Lyme disease in the late stage, stage III).
Reactive arthritis can be associated with other disease manifestations; they are presented in Table 4.26. IN DE-VERSION 4.28?
In the diagnosis of a “reactive arthritis” anamnestic research is to be performed to determine whether there are indications of one of the above-mentioned infections. Accordingly, the following significant anamnestic aspects result:
- Chlamydia trachomatis infection
(with and without symptoms) - Enteritis
- Atypical pneumonia

Additionally, it should be remembered that arthritides also are found in Chlamydophila pneumoniae und Mycoplasma pneumoniae; and that in Chlamydioses, Yersiniosis and also in cases of Mycoplasma pneumoniae, the pathogen has been detected in the synovial fluid or in the articular effusion, respectively.
Using laboratory tests (culture, serology), an existing or previous infection can be detected in 50% of the cases. Other laboratory tests, particularly so-called inflammation markers (BSG, CRP, leukocytosis) are not relevant in cases involving reactive arthritis.
With regard to Chlamydia trachomatis, pathogen detection in a urethral smear or in urine using PCR is appropriate.
Chronic courses, i.e. a duration of illness exceeding 6 months, are observed in nearly 20% of the patients [222].
NSAIDs are used for treatment, but only for pain relief since they have no influence on the disease course or disease duration. In contrast, sulfasalazines [224] and TNF antibodies develop a certain efficacy.
An antibiotic treatment is recommended in cases of acute chlamydiosis with the objective of reducing the frequency of reactive arthritis. However, corresponding studies are not available [223]. In cases of chronic reactive arthritis, the findings on the efficacy of an antibiotic treatment are a matter of controversy [220, 226, 227 - 231].
Overview of the symptomatology and treatment of LD and chronic coinfections
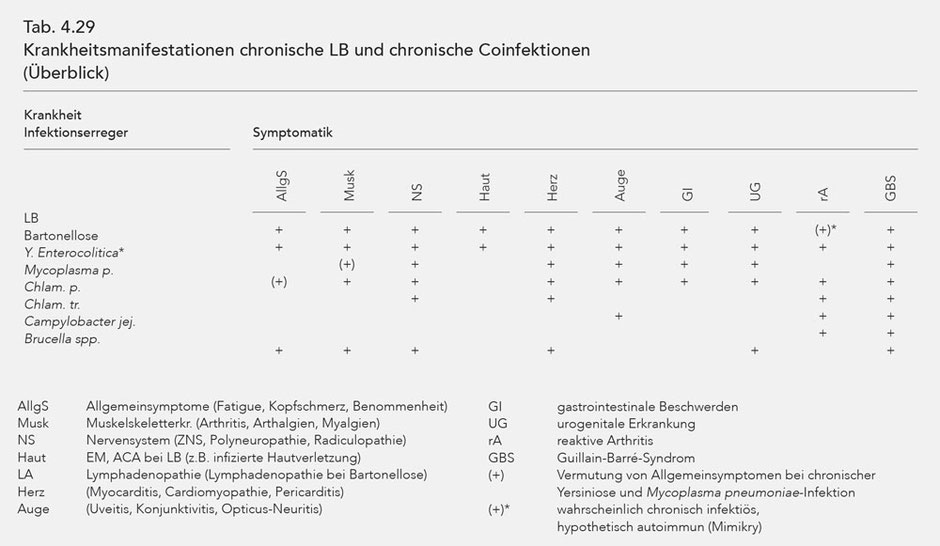
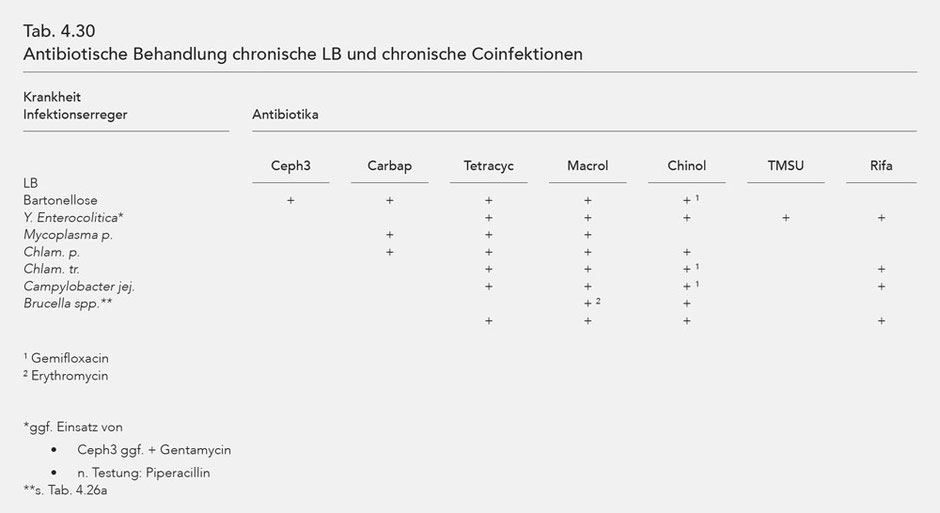
An informative overview of the different disease manifestations of LD and the significant coinfections is given in Table 4.27. The overview shows than there is substantial symptom overlap in cases of LD, bartonellosis, Y. enterocolitica and Mycoplasma pneumoniae. Additionally, Chlamydophila pneumoniae also exhibits some overlap in the symptomatology. Chlamydia trachomatis and Campylobacter jejuni are primarily characterized by reactive arthritis and the rare Guillain-Barré syndrome. Only in cases of chronic Lyme disease does the antibiotic treatment (Table 4.28) involve the use of cephalosporins of the 3rd generation and, if necessary, of carbapenems. Otherwise, the focus is generally on the tetracyclines, the macrolides, to some extent on the quinolones, particularly gemifloxacin; all of which exhibit an intracellular and extracellular efficacy.
Conclusion
At the end of the 20th century, a number of infectious diseases attracted the attention of medical and health policy interests. This is primarily due to the fact these diseases frequently have a chronic course. In Europe and North America, but also in many other areas of the world, these chronic diseases are caused by the following pathogens: Borrelia burgdorferi, Bartonella henselae, Mycoplasma pneumoniae, Chlamydophila pneumoniae, Chlamydia trachomatis, Yersinia enterocolitica. In North America, HGA (Human Granulocytic Anaplasma) and Babesias are also important, whereas infections with these pathogens are a rarity in Europe. In addition, other pathogens discussed in the text are of secondary importance with regard to their frequency of occurrence. Borrelia burgdorferi is transmitted by ticks; in some cases this also applies to Bartonella henselae. The two diseases can also be simultaneously transmitted by tick bites. The remaining pathogens mentioned have other modes of transmission. — Of all the above-mentioned infectious diseases, Lyme borreliosis (Borrelia burgdorferi) is by far the most frequent infectious disease with a chronic course. In addition, this disease (Lyme borreliosis, Lyme disease) is the most thoroughly investigated of all the above-mentioned infectious diseases. On the basis of the special status of Lyme borreliosis, the other infections have been termed “coinfections” in the literature. Lyme borreliosis can be accompanied by one or more coinfections (double or multiple infections). Coinfections exacerbate the expression of the disease and hinder the therapeutic success. The symptomatology of Lyme borreliosis and the so-called coinfections exhibit high degrees of overlap. A subtle diagnostic analysis is required to account for all of the infectious diseases which could (possibly) be present. The diagnostic and therapeutic options in cases of chronic infectious diseases are limited. This applies to Lyme borreliosis and even more for the coinfections. No adequate laboratory methods are available for the important coinfection Bartonella henselae, and there are no official guidelines available with regard to its antibiotic treatment. All of the above-mentioned pathogens are capable of intracellular localization; thus, (with the exception of B. burgdorferi) only intracellularly acting antibiotics are used. Despite this, the failure rate of the antibiotic treatment of the coinfections is high; on the other hand, with particular regard to Bartonella henselae the opinions on an adequate antibiotic therapy are very controversial. Since the clinical and scientific significance of these chronic infectious diseases has meanwhile been recognized, it is now imperative to develop and improve diagnostic and particularly therapeutic measures for the chronic infectious diseases.
- References
-
- Maeda K, Markowitz N, Hawley RC, Ristic M, Cox D, McDade JE. Human infection with Ehrlichia canis, a leukocytic rickettsia. N Engl J Med 1987; 316(14): 853-6.
- Chen SM, Dumler JS, Bakken JS, Walkder DH. Identification of a granulocytotrophic ehrlichia species as the etiologic agent of human disease. J Clin Microbiol 1994; 32(3): 589-95.
- Demma LJ, Holman RC, McQuiston JH, Krebs JW, Swerdlow DL. Epidemiology of human ehrlichiosis and anaplasmosis in the United States, 2001-2001. Am J Trop Med Hyg 2005; 73(2): 400-9.
- Björsdorff A, Wittesjö B, Berglun J, Massung RF, Eliasson I. Human granulocytic ehrlichiosis as a common cause of tick-associated fever in Southeast Sweden: report from a prospective clinical study. Scand J Infect Dis 2002; 34(3): 187-91.
- Stanczak J, Gabre RM, Kruminis-Lozowska W, Racewicz M, Kubica-Biemat B. Ixodes ricinus as a vector of Borrelia burgdorferi sensu lato, Anaplasma phagocytophilum and Babesia microti in urban and suburban forests. Ann Agric Environ Med 2004; 11(1): 109-14.
- Fingerle V, Munderloh UG, Liegl G, Wilske B. Coexistence of ehrlichiae of the phagocytophila group with Borrelia burgdorferi in Ixodes ricinus from Southern Germany. Med Microbiol Immunol 1999; 188(3): 145-9.
- Oehme R, Hartelt K, Backe H, Brockmann S, Kimming P. Foci of tick-borne diseases in southwest Germany. Int J Med Microbiol 2002; 291 Suppl 33:22-9.
- Schouls LM, Van De Pol I, Rijpkema SG, Schot CS. Detection and identification of Ehrlichia, Borrelia burgdorferi sensu lato, and Bartonella species in Dutch Ixodes ricinus ticks. J Clin Microbiol 1999; 37(7): 2215-22.
- Baumgarten B, Harrer TH, Röllinghoff M, Bogdan C. Prevalence of human granulocytic Ehrlichiosis in Ixodes ticks from Southern Germany: Evidence for genetic heterogenety. VIII International Conference on Lyme Borreliosis and other Emerging Tick-Borne Diseases, Munich, 1999 (abstract).
- Hildebrandt A, Schmidt KH, Wilske B, Dom W, Straube E, Fingerle V. Prevalence of four species of Borrelia burgdorferi sensu lato and coinfection with Anaplasma phagocytophila in Ixodes ricinus ticks in central Germay. Eur J Clin Microbiol Infect Dis 2003; 22(6): 364-7.
- Schauber EM, Gertz SJ, Maple WT, Ostfeld RS. Coinfection of blacklegged ticks (Acari: Ixodidae) in Dutchess County, New York, with the agents of Lyme disease and human granulocytic ehrlichiosis. J Med Entomol 1998; 35(5): 901-3.
- Schwartz I, Fish D, Daniels TJ. Prevalence of the rickettsial agent of human granulocytic ehrlichiosis in ticks from a hyperendemic focus of Lyme disease. N Engl J Med1997; 337(1): 49-50.
- Bakken JS, Krueth J, Tilden RL, Dumler JS, Kristiansen BE. Serological evidence of human granulocytic ehrlichiosis in Norway. Eur J Microbiol Infect Dis 1996; 15(10): 829-832.
- Hermanowska-Szpakowicz T, Skotarczak B, Kondrusik M, Rymaszewska A, Sawczuk M, Maciejewska A, Adamska M, Pancewicz S, Zajkowska J. Detecting DNAs of Anaplasma phagocytophilum and Babesia in the blood of patients suspected of Lyme-disease. Ann Agric Envir Med 2004; 11(2): 351-4.
- Pusterla N, Weber R, Wolfensberger C, Schär G, Zbinden R, Fierz W, Madigan JE, Dumler JS, Lutz H.Serological evidence of human granulocytic ehrlichiosis in Switzerland. Eur J Clin Microbiol Infect Dis 1998; 17(3): 207-9.
- Magnarelli LA, Dumler JS, Anderson JF, Johnson RC, Fikring E. Coexistence of antibodies to tick-borne pathogenes of babesiosis, ehrlichiosis, and Lyme borreliosis in human sera. J Clin Microbiol 1995; 33(11): 3054-7.
- Bakken JS, Dumler S. Human granulocytic anaplasmosis. Infect Dis Clin North Am 2008; 22(3): 433-48.
- Aguero-Rosenfeld ME, Horowitz HW, Wormser GP, McKenna DF, Nowakowski J, Muñoz J, Dumler JS. Human granulocytic ehrlichiosis: A case series from a medical center in New York State. Ann Intern Med 1996; 125(11): 904-8.
- Roland WE, McDonald G, Caldwell CW, Everett ED. Ehrlichiosis – a cause of prolonged fever. Clin Infect Dis 1995; 20(4): 821-5.
- Bakken JS, Dumler JS. Clinical diagnosis and treatment of human granulocytotropic anaplasmosis. Ann N Y Acad Sci 2006; 1078: 236-47.
- Skotarczak B, Wodecka B, Cichocka A. Coexistence DNA of Borrelia burgdorferi sensu lato and Babesia microti in Ixodes ricinus ticks from north-western Poland. Ann Agric Environ Med 2002; 9(1): 25-8.
- Skotarczak B, Rymaszewska A, Wodecka B, Sawczuk M. Molecular evidence of coinfection of Borrelia burgdorferi sensu lato, human granulocytic ehrlichiosis agent, and Babesia microti in ticks from northwestern Poland. J Parasitol 2003; 89(1): 194-6.
- Halos L, Jamal T, Maillard R, Beugnet F, Le Menach A, Boulouis HJ, Vayssier-Taussat M. Evidence of Bartonella sp. in questing adult and nymphal Ixodes ricinus ticks from France and co-infection with Borrelia burgdorferi sensu lato and Babesia sp. Vet Res 2005; 36(1): 79-87.
- Schwartz I, Fish D, Daniels TJ. Prevalence of the rickettsial agent of human granulocytic ehrlichiosis in ticks from a hyperendemic focus of Lyme disease. N Engl J Med 1997; 337(1): 49-50.
- Hermanowska-Szpakowicz T, Skotarczak B, Kondrusik M, Rymaszewska A, Sawczuk M, Maciejewska A, Adamska M, Pancewicz S, Zajkowska J. Detecting DNAs of Anaplasma phagocytophilum and Babesia in the blood of patients suspected of Lyme disease. Ann Agric Environ Med 2004; 11(2): 351-4.
- Arnez M, Luznik-Bufon T, Avsic-Zupanc T, Ruzic-Sabljic E, Petrovec M, Lotric-Furlan S, Strle F. Causes of febrile illness after a tick bite in Slovenian children. Pediatr Infect Dis 2003; 22(12): 1078-83.
- Stricker RB, Gaito A, Harris NS, Burrascano JJ. Coinfection in patients with lyme disease: how big a risk? Clin Infect Dis 2003; 37(9): 1277-8.
- Krause PJ, Telford SR 3rd, Spielman A, Sikand V, Ryan R, Christianson D, Burke G, Brassard P, Pollack R, Peck J, Persing DH. Concurrent Lyme disease and babesiosis. JAMA 1996; 275(21): 1657-60.
- Wang TJ, Liang MH, Shangha O, Phillips CB, Lew RA, Wright EA, Berardi V, Fossel AH, Shadick NA. Coexposure to Borrelia burgdorferi and Babesia microti does not worsen the long-term outcome of lyme disease. Clin Infect Dis 2000; 31(5): 1149-54.
- Krause PJ, McKay K, Thompson CA, Sikand VK, Lentz R, Lepore T, Closter L, Christianson D, Telford SR, Persing D, Radolf JD, Spielman A. Deer-Associated Infection Study Group. Disease-specific diagnosis of coinfecting tickborne zoonosis: babesiosis, human granulocytic ehrlichiosis, and Lyme disease. Clin Infect Dis2002; 34(9): 1184-91.
- Hatcher JC, Greenberg PD, Antique J, Jimenez-Lucho VE. Severe babesiosis in Long Island: review of 34 cases and their complications. Clin Infect Dis 2001; 32(8): 1117-25.
- Krause PJ, Telford SR 3rd, Spielman A, Sikand V, Ryan R, Christianson D, Burke G, Brassard P, Pollack R, Peck J, Persing DH. Concurrent Lyme disease and babesiosis. Evidence for increased severity and duration of illness. JAMA 1996; 275(21): 1657-60.
- Reubush TK 2nd, Cassaday PB, Marsh HJ, Lisker SA, Lisker SA, Voorhees DB, Mahoney EB, Healy GR. Human babesiosis on Nantucket Island. Clinical features. Ann Intern Med 1977; 86(1): 6-9.
- White DJ, Talarico J, Chang HG, Birkhead GS, Heimberger T, Morse DL. Human babesiosis in New York State: Review of 139 hospitalized cases and analysis of prognostic factors. Arch Intern Med 1998; 158(19): 2149-54.
- Maeda K, Markowitz N, Hawley RC, Ristic M, Cox D, McDade JE. Human infection with Ehrlichia canis, a leukocytic rickettsia. N Engl J Med 1987; 316(14): 853-6.
- Chen SM, Dumler JS, Bakken JS, Walker DH. Identification of a granulocytotropic Ehrlichia species as the etiologic agent of human disease. J Clin Microbiol 1994; 32(3): 589-95.
- Nadelman RB, Horowitz HW, Hsieh TC, Wu JM, Aguero-Rosenfeld ME, Schwartz I, Nowakowski J, Varde S, Wormser GP. Simultaneous human granulocytic ehrlichiosis and Lyme borreliosis. N Engl J Med 1997; 337(1): 27-30.
- Schwartz I, Fish D, Daniels TJ. Prevalence of the rickettsial agent of human granulocytic ehrlichiosis in ticks from a hyperendemic focus of Lyme disease. N Engl J Med 1997; 337(1): 49-50.
- Lockhart JM, Davidson WR, Stallknecht DE, Dawson JE, Howerth EW. Isolation of Ehrlichia chaffeensis from wild white-tailed deer (Odocoileus virginianus) confirms their role as natural reservoir hosts. J Clin Microbiol 1997; 35(7): 1681-6.
- Kocan AA, Levesque GC, Withworth LC, Murphy GL, Ewing SA, Barker RW. Naturally occurring Ehrlichia chaffeensis infection in coyotes from Oklahoma. Emerg Infect Dis 2000; 6(5): 477-80.
- Horowitz HW, Kilchevsky E, Haber S, Aguero-Rosenfeld M, Kranwinkel R, James EK, Wong SJ, Chu F, Liveris D, Schwartz I. Perinatal transmission of the agent of human granulocytic ehrlichiosis. N Engl Med 1998; 339(6): 375-8.
- Bakken JS, Dumler S. Human granulocytic anaplasmosis. Infect Dis Clin North Am 2008; 22(3): 433-48.
- Bakken JS, Krueth JK, Lund T, Malkovitch D, Asanovich K, Dumler JS. Exposure to deer blood may be a cause of human granulocytic ehrlichiosis. Clin Infect Dis 1996; 23(1): 198.
- Krause PJ, Wormser GP. Nosocomial transmission of human granulocytic anaplasmosis? JAMA 2008; 300(19): 2308-9.
- Dumler JS, Bakken JS. Ehrlichial diseases of humans: emerging tick-borne infections. Clin Infect Dis 1995; 20(5): 1102-10.
- Dawson JE, Fishbein DB, Eng TR, Redus MA, Green NR. Diagnosis of human ehrlichiosis with the indirect fluorescent antibody test: kinetics and specificity. J Infect Dis 1990; 162(1): 91-5.
- Brouqui P, Bacellar F, Baranton G, Birtles RJ, Bjoërsdorff A, Blanco JR, Caruso G, Cinco M, Fournier PE, Francavilla E, Jensenius M, Kazar J, Laferl H, Lakos A, Lotric Furlan S, Maurin M, Oteo JA, Parola P, Perez-Eid C, Peter O, Postic D, Raoult D, Tellez A, Tselentis Y, Wilske B; ESCMID Study Group on Coxiella, Anaplasma, Rickettsia and Bartonella; European Network for Surveillance of Tick-Borne Diseases.Guidelines for the diagnosis of tick-borne bacterial diseases in Europe. Clin Microbiol Infect 2004; 10(12): 1108-32.
- Bakken JS, Haller I, Riddell D, Walls JJ, Dumler JS. The serological response of patients infected with the agent of human granulocytic ehrlichiosis. Clin Infect Dis 2002; 34(1): 22-7.
- Bakken JS, Dumler JS. Ehrlichiosis and anaplasmosis. Infect Med 2004; 21:433.
- Paddock CD, Childs JE. Ehrlichia chaffeensis: a prototypical emerging pathogen. Clin Microbiol Rev 2003; 16(1): 37-64.
- Bakken JS, Krueth J, Wilson-Nordskog C, Tilden RL, Asanovich K, Dumler JS. Clinical and laboratory characteristics of human granulocytic ehrlichiosis. JAMA 1996; 275(3): 199-205.
- Bakken JS, Aguero-Rosenfeld ME, Tilden RL, Wormser GP, Horowitz HW, Raffalli JT, Baluch M, Riddell D, Walls JJ, Dumler JS. Serial measurements of hematologic counts during the active phase of human granulocytic ehrlichiosis. Clin Infect Dis 2001; 32(6): 862-70.
- Chapman AS, Bakken JS, Folk SM, Paddock CD, Bloch KC, Krusell A, Sexton DJ, Buckingham SC, Marshall GS, Storch GA, Dasch GA, McQuiston JH, Swerdlow DL, Dumler SJ, Nicholson WL, Walker DH, Eremeeva ME, Ohl CA; Tickborne Rickettsial Diseases Working Group; CDC. Diagnosis and management of tickborne rickettsial diseases: Rocky Mountain spotted fever, ehrlichiosis, and anaplasmosis - - United States: a pracitcal guide for physicians and other health-care and public health professionals. MMWR Recomm Rep 2006; 55(RR-4): 1-27.
- Standaert SM, Yu T, Scott MA, Childs JE, Paddock CD, Nicholson WL, Singleton J Jr, Blaser MJ. Primary isolation of Ehrlichia chaffeensis from patients with febrile illnesses: clinical and molecular characteristics. J Infect Dis 2000; 181(3): 1082-8.
- Felek S, Unver A, Stich RW, Rikihisa Y. Sensitive detection of Ehrlichia chaffeensis in cell culture, blood, and tick specimens by reverse transcription-PCR. J Clin Microbiol 2001; 39(2): 460-3.
- Walls JJ, Caturegli P, Bakken JS, Asanovich KM, Dumler JS. Improved sensitivity of PCR for diagnosis of human granulocytic ehrlichiosis using epank1 genes of Ehrlichia phagocytophila-group ehrlichiae. J Clin Microbiol 2000; 8(1): 354-6.
- Vannier E, Gewurz BE, Krause PJ. Human babesiosis. Infect Dis Clin North Am 2008; 22(3): 469-88.
- Zintl A, Mulcahy G, Skerrett HE, Taylor SM, Gray JS. Babesia divergens, a bovine blood parasite of veterinary and zoonotic importance. Clin Microbiol Rev 2003; 16(4): 622-36.
- Leiby DA. Babesiosis and blood transfusion: flying under the radar. Vox Sang 2006; 90(3): 157-65.
- Fox LM, Wingerter S, Ahmed A, Arnold A, Chou J, Rhein L, Levy Neonatal babesiosis: case report and review of the literature. Pediatr Infect Dis J 2006; 25(2): 169-73.
- Sethi S, Alcid D, Kesarwala H, Tolan RW Jr. Probable congenital babesiosis in infant, new jersey, USA. Emerg Infect Dis 2009; 15(5): 788-91.
- Vannier E, Gewurz BE, Krause PJ. Human babesiosis, Infect Dis Clin North Am, 22:469, 2008
- Krause PJ, Ryan R, Telford S 3rd, Persing D, Spielman A. Efficacy of immunoglogulin M serodiagnostic test for rapid diagnosis of acute babesiosis. J Clin Microbiol 1996; 34(8): 2014-6.
- Krause PJ, Telford SR 3rd, Ryan R, Conrad PA, Wilson M, Thomford JW, Spielman A. Diagnosis of babesiosis: evaluation of a serologic test for the detection of Babesia microti antibody. J Infect Dis 1994; 169(4): 923-6.
- Ruebush TK 2nd, Chisholm ES, Sulzer AJ, Healy GR. Development and persistence of antibody in persons infected with Babesia microti. Am J Trop Med Hyg 1981; 30(1): 291-2.
- Bass JW, Vincent JM, Person DA. The expanding spectrum of Bartonella infections: II. Cat-scratch disease. Pediatr Infect Dis J 1997; 16(2): 163-79.
- Spach DH, Koehler JE. Bartonella-associated infections. Infect Dis Clin North Am 1998; 12(1):137-55.
- Wear DJ, Margileth AM, Hadfield TL, Fischer GW, Schlagel CJ, King FM. Cat scratch disease: A bacterial infection. Science 1983; 221(4618): 1403-5.
- Englisch CK, Wear DJ, Margileth AM, Lissner CR, Walsh GP. Cat-scratch disease. Isolation and culture of the bacterial agent. JAMA 1988; 259(9): 1347-52.
- Bonatti M, Mendez J, Guerrero I, Krishna M, Ananda-Michel J, Yao J, Steers JL, Hellinger W, Dickson RC, Alvarez S. Disseminated Bartonella infection following liver transplantation. Transpl Int 2006; 19(8): 683-7.
- Thudi KR, Kreikemeier TJ, Phillips NJ, Salvalaggio PR, Kennedy DJ, Hayashi PH. Cat scratch disease causing hepatic masses after liver transplant. Liver Int 2007; 27(1): 145-8.
- Bhatti Z, Berenson CS. Adult systemic cat scratch disease associated with therapy for hepatitis C. BMC Infect Dis 2007; 7:8.
- Koehler JE, Glaser CA, Tappero JW. Rochalimaea henselae infection. A new zoonosis with the domestic cat as reservoir. JAMA 1994; 271(7): 531-5.
- Chomel BB, Abbott RC, Kasten RW, Floyd-Hawkins KA, Kass PH, Glaser CA, Pedersen NC, Koehler JE. Bartonella henselae prevalence in domestic cats in California: risk factors and association beween bacteremia and antibody titers. J Clin Microbiol 1995; 33(9): 2445-50.
- Moriarty RA, Margileth AM. Cat-scratch disease. Infect Dis Clin North Am 1987; 1(3): 575-90.
- Cunningham ET, Koehler JE. Ocular bartonellosis. Am J Ophthalmol 2000; 130(3): 340-9.
- Bhatti MT, Asif R, Bhatti LB. Macular star in neuroretinitis. Arch Neurol 2001; 58(6): 1008-9.
- Suhler EB, Lauer AK, Rosenbaum JJ. Prevalence of serologic evidence of cat scratch disease in patients with neuroretinitis. Ophthalmology 2000; 107(5): 871-6.
- Reed JB, Scales DK, Wong MT, Lattuada CP Jr, Dolan MJ, Schwab IR. Bartonella henselae neuroretinitis in cat scratch disease. Diagnosis, management, and sequelae. Ophthalmology 1998; 105(3): 459-66.
- Marra CM. Neurologic complications of Bartonella henselae infection. Curr Opin Neurol 1995; 8(3): 164-9.
- Selby G, Walker GL. Cerebral arteritis in cat-scratch disease. Neurology 1979; 29(10): 1413-8.
- Baylor P, Garoufi A, Karpathios T, Lutz J, Mogelof J, Moseley D. Transverse myelitis in 2 patients with Bartonella henselae infection (cat scratch disease). Clin Infect Dis 2007; 45(4): e42-5.
- Maman E, Bickels J, Ephros M, Paran D, Comaneshter D, Metzkor-Cotter E, Avidor B, Varon-Graidy M, Wientroub S, Giladi M. Musculoskeletal manifestations of cat scratch disease, Clin Infect Dis 2007; 45(12): 1535-40.
- Giladi M, Maman E, Paran D, Bickels J, Comaneshter D, Avidor B, Varon-Graidy M, Ephros M, Wientroub S. Cat-scratch disease-associated arthropathy. Arthritis Rheum 2005; 52(11): 3611-7.
- Ben-Ami R, Ephros M, Avidor B, Katchman E, Varon M, Leibowitz C, Comaneshter D, Giladi M. Cat-scratch disease in elderly patients. Clin Infect Dis 2005; 41(7): 969-74.
- Spach DH, Kaplan SL. Microbiology, epidemiology, clinical manifestations and diagnosis of cat scratch disease. UpToDate, 2008.
- Jensen WA, Fall MZ, Rooney J, Kordick DL, Breitschwerdt EB. Rapid identification and differentiation of Bartonella species using a single-step PCR assay. J Clin Microbiol 2000; 38(5): 1717-22.
- Ridder GJ, Boedeker CC, Technau-Ihling K, Grunow R, Sander A. Role of cat-scratch disease in lymphadenopathy in the head and neck. Clin Infect Dis 2002; 35(6): 643-9.
- Maman E, Bickels JS, Ephros M, Paran D, Comaneshter D, Metzkor-Cotter E, Avidor B, Varon-Graidy M, Wientroub S, Giladi M. Musculoskeletal manifestations of cat scratch disease. Clin Infect Dis 2007; 45(12): 1535-40.
- Gilaldi M, Maman E, Paran D, Bickels J, Comaneshter D, Avidor B, Varon-Graidy M, Ephros M, Wientroub S. Cat-scratch disease-associated arthropathy. Arthritis Rheum 2005; 52(11):3611-7.
- Ridder GJ, Boedeker CC, Technau-Ihling K. Cat-scratch disease: Otolaryngologic manifestations and management. Otolaryngol Head Neck Surg 2005; 132(3):353-8.
- Tsujino K, Tsukahara M, Tsuneoka H, Ichihara K, Furuya T, Kawauchi S, Oga A, Sasaki K. Clinical implication of prolonged fever in children with cat scratch disease. J Infect Chemother 2004; 10(4):227-33.
- Methkor-Cotter E, Kletter Y, Avidor B, Varon M, Golan Y, Ephros M, Giladi M. Long-term serological analysis and clinical follow-up of patients with cat scratch disease. Clin Infect Dis 2003; 37(9):1149-54.
- Murakami K, Tsukahara M, Tsuneoka H, Iino H, Ishida C, Tsujino K, Umeda A, Furuya T, Kawauchi S, Sasaki K. Cat scratch disease: analysis of 130 seropositive cases. J Infect Chemother 2002; 8(4): 349-52.
- Eskow E, Rao RV, Mordechai E. Concurrent infection of the central nervous system by Borrelia burgdorferi and Bartonella henselae: evidence for a novel tick-borne disease complex. Arch Neurol 2001; 58(9):1357-63.
- Dietrich F, Schmidgen T, Maggi RG, Richter D, Matuschka FR, Vonthein R, Breitschwerdt EB, Kempf VAJ. Prevalence of Bartonella henselae and Borrelia burgdorferi sensu lato DNA in ixodes ricinus ticks in Europe. Appl Environ Microbiol 2010; 76(5): 1395-8.
- Windsor JJ. Cat-scratch disease: epidemiology, aetiology and treatment. Br J Biomed Sci 2001; 58(2): 101-10.
- Guptill L. Bartonellosis. Vet Microbiol 2010; 140(3-4): 357-59.
- Cotell SL, Noskin GA. Bacillary angiomatosis. Clinical and histologic features, diagnosis, and treatment. Arch Intern Med 1994; 154(5): 524-8.
- Pulliainen AT, Dehio C. Bartonella henselae: subversion of vascular endothelial cell functions by translocated bacterial effector proteins. Int J Biochem Cell Biol 2009; 41(3): 507-10.
- Schaller DJ. Bartonella Diagnosis and Treatment. Hope Academic Press, Tampa, Florida, 2008.
- Riess T, Andersson SG, Lupus A, Schaller M, Schäfer A, Kyme P, Martin J, Wälzlein JH, Ehehalt U, Lindroos H, Schirle M, Nordheim A, Autenrieth IB, Kempf VA Bartonella adhesin mediates a proangiogenic host cell response. J Exp Med 2004; 200(10): 1267-78.
- Dehio C, Meyer M, Berger J, Schwarz H, Lanz C. Interaction of Bartonella henselae with endothelial cells results in bacterial aggregation on the cell surface and the subsequent engulfment and internalisation of the bacterial aggregate by a unique structure, the invasome. J Cell Sci 1997; 100(Pt 18): 2141-54.
- Kempf VA, Lebiedziejwski M, Alitalo K, Wälzlein JH, Ehehalt U, Fiebig J, Huber S, Schütt B, Sander CA, Müller S, Grassl G, Yazdi AS, Brehm B, Autenrieth IB. Activation of hypoxia-inducible factor-1 in bacillary angiomatosis: evidence for a role of hypoxia-inducible factor-1 in bacterial infections. Circulation 2005; 111(8): 1054-62.
- Kirby JE. In vitro model of Bartonella henselae-induced angiogenesis. Infect Immun 2004; 72(12): 7315-7.
- Dehio C. Recent progress in understanding Bartonella-induced vascular proliferation. Curr Opin Microbiol 2003; 6(1): 61-5.
- Resto-Ruiz S, Burgess A, Anderson BE. The role of the host immune response in pathogenesis of Bartonella henselae. DNA Cell Biol 2003; 22(6): 431-40.
- Kempf VA, Volkmann B, Schaller M. Sander CA, Alitalo K, Riess T, Autenrieth IB. Evidence of a leading role for VEGF in Bartonella henselae-induced endothelial cell proliferations. Cell Microbiol 2001; 3(9): 623-32.
- Mernaugh G, Ihler GM. Deformation factor: an extracellular protein synthesized by Baronella bacilliformis that deforms erythrocyte membranes. Infect Immun 1992; 60(3): 937-43.
- Rolain JM, Amoux D, Parzy D, Sampol J, Raoult D. Experimental infection of human erythrocytes from alcoholic patients with Bartonella Quintana. Ann NY Acad Sci 2003; 990: 605-11.
- Rolain JM, Foucault C, Guieu R, La Scola B, Brouqui P, Raoult D. Bartonella Quintana in human erythrocytes. Lancet 2002; 360(9328): 226-8.
- Mursic VP, Wanner G, Reinhardt S, Wilske B, Busch U, Marget W. Formation and cultivation of Borrelia burgdorferi spheroplast-L-form variants. Infection 1996; 24(3): 218-26.
- Liu NY. Randomized trial of doxycycline vs. amoxicillin/probenecid for the treatment of Lyme arthritis: treatment of non responders with iv penicillin or ceftriaxone. Arthritis Rheum 1989; 32: 46.
- Kraiczy P, Skerka C, Kirschfink M, Zipfel PF, Brade V. Immune evasion of Borrelia burgdorferi: insufficient killing of the pathogens by complement and antibody. Int J Med Microbiol 2002; 291 Suppl 33: 141-146.
- Kraiczy P, Skerka C, Zipfel PF, Brade V. Complement regulator-acquiring surface proteins of Borrelia burgdorferi: a new protein family involved in complement resistance. Wien Klin Wochenschr 2002; 114(13-14): 568-573.
- Mursic VP, Wanner G, Reinhardt S, Wilske B, Busch U, Marget W. Formation and cultivation of Borrelia burgdorferi spheroplast-L-form variants. Infection 1996; 24(3): 218-26.
- Brorson O, Brorson SH. An in vitro study of the susceptibility of mobile and cystic forms of Borrelia burgdorferi to hydroxychloroquine. Int Microbiol 2002; 5(1): 25-31.
- Bass JW, Freitas BC, Freitas AD, Sisler CL, Chan DS, Vincent JM, Person DA, Claybaugh JR, Wittler RR, Weisse ME, Regnery RL, Slater LN. Prospective randomized double blind placebo-controlled evaluation of azithromycin for treatment of cat-scratch disease. Pediatr Infect Dis J 1998; 17(6): 447-52.
- Chia JK, Nakata MM, Lami JL, Park SS, Ding JC. Azithromycin for the treatment of cat-scratch disease. Clin Infect Dis 1998; 26(1): 193-4.
- Margileth AM. Antibiotic therapy for cat-scratch disease: clinical study of therapeutic outcome in 268 patients and a review of the literature. Pediatr Infect Dis J 1992; 11(6): 474-8.
- Bogue CW, Wise JD, Gray GF, Edwards KM. Antibiotic therapy for cat-scratch disease? JAMA 1989; 262(6): 813-6.
- Arisoy ES, Correa AG, Wagner ML, Kaplan SL. Hepatosplenic cat-scratch disease in children: selected clinical features and treatment. Clin Infect Dis 1999; 28(4): 778-84.
- Spach DH, Kanter AS, Dougherty M, Larson AM, Coyle MB, Brenner DJ, Swaminathan B, Matar GM, Welch DF, Root RK, et al. Bartonella (Rochalimaea) quintana bacteremia in inner-city patients with chronic alcoholism. N Engl J Med 1995; 332(7): 424-8.
- Foucault C, Raoult D, Brouqui P. Randomized open trial of gentamicin and doxycycline for eradication of Bartonella quintana from blood in patients with chronic bacteremia. Antimicrob Agents Chemother 2003; 47(7): 2204-7.
- Raoult D, Fournier PE, Vandenesch F, Outcome and treatment of Bartonella endocarditis. Outcome and treatment of Bartonella endocarditis. Arch Intern Med 2003; 163(2): 226-30.
- Rolain JM, Brouqui P, Koehler JE, Maguina C, Dolan MJ, Raoult D. Recommendations for treatment of human infections caused by Bartonella species. Antimicrob Agents Chemother 2004; 48(6): 1921-33.
- Foucault C, Brouqui P, Raoult D. Bartonella quintana characteristics and clinical management. Emerg Infect Dis 2006; 12(2): 217-23.
- Relman DA. Has trench fever returned? N Engl J Med 1995; 332(7): 463-4.
- Vinson JW. In vitro cultivation of the rickettsial agent of trench fever. Bull World Health Organ 1966; 35(2): 155-64.
- Rolain JM, Foucault C, Guieu R, La Scola B, Brouqui P, Raoult D. Bartonella quintana in human erythrocytes. Lancet 2002; 360(9328): 226-8.
- Stamm WE, Jones RB, Batteiger BE. Introduction to Chlamydial diseases. In: Principles and Practice of Infectious Diseases. 6th ed. Mandell, GL, Bennett, JE, Dolin, R, (Eds), Churchill Livingstone, Philadelphia, PA 2005, p. 2236.
- Jackson LA, Grayston JT. Chlamydia pneumoniae. In Principles and Practice of Infectious Diseases. Madell, GL, Bennett, JE, Dolin, R (Eds), 5th ed, Churchill Livingstone, Philadelphia 2000, p. 2007.
- Myhra W, Mordhorst CH, Wang SP, Grayston JT. Clinical features of Chlamydia pneumoniae, strain TWAR, infection in Denmark 1975-1987. In: Chlamydial Infections. Bowie, WR, Caldwell, HD, Jones, RP (Eds), 1980, p. 422.
- Bourke SJ, Lightfoot NF. Chlamydia pneumoniae: defining the clinical spectrum of infection requires precise laboratory diagnosis. Thorax 1995; 50 Suppl 1: S43-8.
- Tuuminen T, Palomäki P, Paavonen J. The use of serologic tests for the diagnosis of chlamydial infections. J Microbiol Methods 2000; 42(3): 265-79.
- Kumar S, Hammerschlag MR. Acute respiratory infection due to Chlamydia pneumoniae: current status of diagnostic methods. Clin Infect Dis 2007; 44(4): 568-76.
- Gaydos CA, Roblin PM, Hammerschlag MR, Hyman CL, Eiden JJ, Schachter J, Quinn TC. Diagnostic utility of PCR-enzyme immunoassay, culture, and serology for detection of Chlamydia pneumoniae in symptomatic and asymptomatic patients. J Clin Microbiol 1994; 32(4): 903-5.
- Vergis EN, Indorf A, File TM Jr, Phillips J, Bates J, Tan J, Sarosi GA, Grayston JT, Summersgill J, YU VL. Azithromycin vs cefuroxime plus erythromycin for empirical treatment of community-acquired pneumonia in hospitalized patients: a prospective, randomized, multicenter trial. Arch Intern Med 2000; 160(9): 1294-300.
- Stamm WE. Chlamydia trachomatis infections: progress and problems. J Infect Dis 1999; 179 Suppl 2: S380-3.
- Centers for Disease Control and Prevention, Workowski KA, Berman SM. Sexually transmitted diseases treatment guidelines, 2006. MMWR Recomm Rep 2006; 55(RR-11): 1-94.
- Quinn TC, Welsh L, Lentz A, Crotchfelt K, Zenilman J, Newhall J, Gaydos C. Diagnosis by AMPLICOR PCR of Chlamydia trachomatis infection in urine samples from women and men attending sexually transmitted disease clinics. J Clin Microbiol 1996; 34(6): 1401-6.
- Chernesky MA, Lee H, Schachter, Burczak JD, Stamm WE, McCormack WM, Quinn TC. Diagnosis of Chlamydia trachomatis urethral infection in symptomatic and asymptomatic men by testing first-void urine in a ligase chainreaction assay. J Infect Dis 1994; 170(5): 1308-11.
- Cook RL, Hutchinson SL. Ostergaard L, Braithwaite RS, Ness RB. Systematic review: noninvasive testing for Chlamydia trachomatis and Neisseria gonorrhea. Ann Intern Med 2005; 142(11): 914-25.
- Keat A. Extra-genital Chlamydia trachomatis infection as sexually-acquired reactive arthritis. J Infect 1992; 25 Suppl 1: 47-9.
- Mollaret HH. Fifteen centuries of Yersiniosis. Contrib Microbiol Immunol 1995; 13: 1-4.
- Ostroff S. Yersinia as an emerging infection: epidemiologic aspects of Yersiniosis. Contrib Microbiol Immunol 1995; 13: 5-10.
- Portnoy DA, Martinez RJ. Role of a plasmid in the pathogenicity of Yersinia species. Curr Top Microbiol Immunol 1985; 118: 29-51.
- Iriarte M, Sory MP, Boland A, Boyd AP, Mills SD, Lambermont I, Cornelis GR. TyeA, a protein involved in control of Yop release and in translocation of Yersinia Yop effectors. EMBO J 1998; 17(7): 1907-18.
- Sarker MR, Sory MP, Boyd AP, Iriarte M, Comelis GR. LcrG is required for efficient translocation of Yersinia Yop effector proteins into eukaryotic cells. Infect Immun 1998; 66(6): 2976-9.
- Boland A, Cornelis GR. Role of YopP in suppression of tumor necrosis factor alpha release by macrophages during Yersinia infection. Infect Immun 1998; 66(5): 1878-84.
- Tacket CO, Narain JP, Sattin R, Lofgren JP, Konigsberg C Jr, Rendtorff RC, Rausa A, Davis BR, Cohen ML. A multistate outbreak of infections caused by Yersinia enterocolitica transmitted by pasteurized milk. JAMA 1984; 251(4): 483-6.
- Ostroff SM, Kapperud G, Lassen J, Aasen S, Tauxe RV. Clinical features of sporadic Yersinia enterocolitica infections in Norway. J Infect Dis 1992; 166(4): 812-7.
- Tauxe RV, Vandepitte J, Wauters G, Martin SM, Goossens V, De Mol P, Van Noyen R, Thiers G. Yersinia enterocolitica infections and pork: the missing link. Lancet 1987; 1(8542): 1129-32.
- Tacket CO, Davis BR, Carter GP, Randolph JF, Cohen ML. Yersinia enterocolitica pharyngitis. Ann Intern Med 1983; 99(1): 40-2.
- Cover TL, Aber RC. Yersinia enterocolitica. N Enl J Med 1989; 321(1): 16-24.
- Black RE, Slome S. Yersinia enterocolitica. Inefct Dis Clin North Am 1988; 2(3): 625-41.
- Reed RP, Robins-Browne RM, Williams ML. Yersinia enterocolitica peritonitis. Clin Infect Dis 1997; 25(6): 1468-9.
- Blinkhorn RJ Jr, Marino JA. Lateral pharyngeal abscess due to Yersinia enterocolitica. Am J Med 1988; 85(6): 851-2.
- Krogstad P, Mendelman PM, Miller VL, Clausen C, Abbott S, Weagant S, Wilson CL, Lewis DB. Clinical and microbiologic characteristics of cutaneuous infection with Yersinia enterocolitica. J Infect Dis 1992; 165(4): 740-3.
- Crowe M, Ashford K, Ispahani P. Clinical features and antibiotic treatment of septic arthritis and osteomyelitis due to Yersinia enterocolitica. J Infect Microbiol 1996; 45(4): 302-9.
- Kellogg CM, Tarakji EA, Smith M, Brown PD. Bacteremia and suppurative lymphadenitis due to Yersinia enterocolitica in a neutropenic patient who prepared chitterlings. Clin Infect Dis 1995; 21(1): 236-7.
- van der Heijden IM, Res PC, Wilbrink B, Leow A, Breedveld FC, Heesemann J, Tak PP. Yersinia enterocolitica: a cause of chronic polyarthritis. Clin Infect Dis 1997; 25(4): 831-7.
- Granfors K, Jalkanen S, von Essen R, Lahesmaa-Rantala R, Isomäki O, Pekkola-Heino K, Merilahti-Palo R, Saario R, Isomäki H, Toivanen A. Yersinia antigens in synovial-fluid cells from patients with reactive arthritis. N Engl J Med 1989; 320(4): 216-21.
- Granfors K, Merilahti-Palo R, Luukkainen R, Möttönen T, Lahesmaa R, Probst P, Märker-Hermann E, Toivanen P. Persistence of Yersinia antigens in peripheral blood cells from patients with Yersinia enterocolitica O:3 infection with or without reactive arthritis. Arthritis Rheum 1998; 41(5): 855-62.
- Leirisalo-Repo M, Suoranta H. Ten-year follow-up study of patient with Yersinia arthritis. Arthritis Rheum 1988; 31(4): 533-7.
- Bottone EJ. Yersinia enterocolitica: the charisma continues. Clin Microbiol Rev 1997; 10(2): 257-76.
- Mäki-Ikola O, Heesemann J, Toivanen A, Granfors K. High frequency of Yersinia antibodies in healthy populations in Finland and Germany. Rheumatol Int 1997; 16(6): 227-9.
- Ostroff SM. Clinical features and diagnosis of Yersinia enterocolitica and Yersinia pseudotuberculosis infection. UpToDate, 2009.
- Lindblom A , Isa A, Norbeck O, Wolf S, Johansson B, Broliden K, Tolfvenstam T. Slow clearance of human parvovirus B19 viremia following acute infection. Clin Infect Dis 2005; 41(8): 1201-3.
- Lowry SM, Brent LH, Menaldino S, Kerr JR. A case of persistent parvovirus B19 infection with bilateral cartilaginous and ligamentous damage to the wrists. Clin Infect Dis 2005; 41(4):e42-4.
- Nikkari S, Roivainen A, Hannonen P, Möttönen T, Luukkainen R, Yli-Jama T, Toivanen P. Persistence of parvovirus B19 in synovial fluid and bone marrow. Ann Rheum Dis 1995; 54(7): 597-600.
- Kühl U, Pauschinger M, Seeberg B, Lassner D, Noutsias M, Poller W, Schultheiss HP. Viral persistence in the myocardium is associated with progressive cardiac dysfunction. Circulation 2005; 112(13): 1965-70.
- Tschöpe C, Bock CT, Kasner M, Noutsias M, Westermann D, Schwimmbeck PL, Pauschinger M, Poller WC, Kühl U, Kandolf R, Schultheiss HP.High prevalence of cardiac parvovirus B19 infection in patients with isolated left ventricular diastolic dysfunction. Circulation 2005; 111(7): 879-86.
- Donoso Mantke O, Nitsche A, Meyer R, Klingel K, Niedrig M. Analysing myocardial tissue from explanted hearts of heart transplant recipients and multi-organ donors for the presence of parvovirus B19 DNA. J Clin Virol 2004; 31(1): 32-9.
- Schenk T, Enders M, Pollak S, Hahn R, Huzly D. High prevalence of human parvovirus B19 DNA in myocardial autopsy samples from subjects without myocarditis or dilative cardiomyopathy. J Clin Microbiol 2009; 47(1): 106-10.
- Eskow E, Rao RV, Mordechai E. Concurrent infection of the central nervous system by Borrelia burgdorferi and Bartonella henselae: evidence for a novel tick-borne disease complex. Arch Neurol 2001; 58(9): 1357-63.
- Grab DJ, Nyarko E, Barat NC, Nikolskaia OV, Dumler JS. Anaplasma phagocytophilum-Borrelia burgdorferi coinfection enhances chemokine, cytokine, and matrix metalloprotease expression by human brain microvascular endothelial cells. Clin Vaccine Immunol 2007; 14(11): 1420-4.
- Mitchell PD, Reed KD, Hofkes JM. Immunoserologic evidence of coinfection with Borrelia burgdorferi, Babesia microti, and human granulocytic Ehrlichia species in residents of Wisconsin and Minnesota. J Clin Microbiol 1996; 34(3):724-7.
- Oleson CV, Sivalingam JJ, O’Neill BJ, Staas WE Jr. Transverse myelitis secondary to coexistent Lyme disease and babesiosis. J Spinal Cord Med 2003; 26(2): 168-71.
- Owen DC. Is Lyme disease always poly microbial?--The jigsaw hypothesis. Med Hypotheses 2006; 67(4): 860-4.
- Swanson SJ, Neitzel D, Reed KD, Belongia EA. Coinfections acquired from ixodes ticks. Clin Microbiol Rev 2006; 19(4): 708-27.
- homas V, Anguita J, Barthold SW, Fikrig E. Coinfection with Borrelia burgdorferi and the agent of human granulocytic ehrlichiosis alters murine immune responses, pathogen burden, and severity of Lyme arthritis. Infect Immun 2001; 69(5): 3359-71.
- Zeidner NS, Dolan MC, Massung R, Piesman J, Fish D. Coinfection with Borrelia burgdorferi and the agent of human granulocytic ehrlichiosis suppresses IL-2 and IFN gamma production and promotes an IL-4 response in C3H/HeJ mice. Parasite Immunol 2000; 22(11): 581-8.
- Wormser GP, Nadelman RB, Dattwyler RJ, Dennis DT, Shapiro ED, Steere AC, Rush TJ, Rahn DW, Coyle PK, Persing DH, Fish D, Luft BJ. Practice guidelines for the treatment of Lyme disease. The Infectious Diseases Society of America. Clin Infect Dis 2000; 31 Suppl 1: 1-14.
- Kristoferitsch W, Stanek G, Kunz C. [Double infection with early summer meningoencephalitis virus and Borrelia burgdorferi]. [Article in German]. Dtsch Med Wochenschr 1986; 111(22): 861-4.
- Hunfeld K-P. Granulocytic Ehrlichia, Babesia, and spotted fever Rickettsia. Not yet widely known tick-borne pathogens of considerable concern for humans at risk in Europe. Biotest Bulletin 2002; 6: 321-344.
- Cadavid D, O’Neill T, Schaefer H, Pachner AR. Localization of Borrelia burgdorferi in the nervous system and other organs in a nonhuman primate model of Lyme disease. Lab Invest 2000; 80(7): 1043-54.
- Wormser GP, Dattwyler RJ, Shapiro ED, Halperin JJ, Steere AC, Klempner MS, Krause PJ, Bakken JS, Strle F, Stanek G, Bockenstedt L, Fish D, Dumler JS, Nadelman RB. The clinical assessment, treatment, and prevention of lyme disease, human granulocytic anaplasmosis, and babesiosis: clinical practice guidelines by the Infectious Diseases Societey of America. Clin Infect Dis 2006; 43(9): 1089-134.
- Krause PJ, Telford SR 3rd, Spielman A, Sikand V, Ryan R, Christianson D, Burke G, Brassard P, Pollack R, Peck J, Persing DH. Concurrent Lyme disease and babesiosis. Evidence for increased severity and duration of illness. JAMA 1996; 275(21): 1657-60.
- Straubinger RK, Straubinger AF, Summers BA, Jacobson RH. Status of Borrelia burgdorferi infection after antibiotic treatment and the effects of corticosteroids: An experimental study. J Infect Dis 2000; 181(3) : 1069-81.
- Stricker RB. Counterpoint: long-term antibiotic therapy improves persistent symptoms associated with lyme disease. Clin Infect Dis 2007; 45(2): 149-57.
- Grab DJ, Nyarko E, Barat NC, Nikolskaia OV, Dumler JS. Anaplasma phagocytophilum-Borrelia burgdorferi coinfection enhances chemokine, cytokine, and matrix metalloprotease expression by human brain microvascular endothelial cells. Clin Vaccine Immunol 2007; 14(11): 1420-4.
- Levin ML, Fish D. Acquisition of coinfection and simultaneous transmission of Borrelia burgdorferi and Ehrlichia phagocytophila by Ixodes scapularis ticks. Infect Immun 2000; 68(4): 2183-6.
- Kocan AA, Levesque GC, Whitworth LC, Murphy GL, Ewing SA, Barker RW. Naturally occurring Ehrlichia chaffeensis infection in coyotes from Oklahoma. Emerg Infect Dis 2000; 6(5): 477-80.
- Goodman JL, Nelson C, Vitale B, Madigan JE, Dumler JS, Kurtti TJ, Munderloh UG. Direct cultivation of the causative agent of human granulocytic ehrlichiosis. N Engl J Med 1996; 334(4): 209-15.
- Meer-Scherrer L, Adelson M, Mordechai E, Lottaz B, Tilton R. Babesia microti infection in Europe. Curr Microbiol 1996; 48(6): 435-7.
- Mitchell PD, Reed KD, Hofkes JM. Immunoserologic evidence of coinfection with Borrelia burgdorferi, Babesia microti, and human granulocytic Ehrlichia species in residents of Wisconsin and Minnesota. J Clin Microbiol 1996; 34(3): 724-7.
- Oleson CV, Sivalingam JJ, O’Neill BJ, Staas WE Jr. Transverse myelitis secondary to coexistent Lyme disease and babesiosis. J Spinal Cord Med 2003; 26(2): 168-71.
- Shoemaker RC, Hudnell HK, House DE, Van Kempen A, Pakes GE, COL40155 Study Team. Atovaquone plus cholestyramine in patients coinfected with Babesia microti and Borrelia burgdorferi refractory to other treatment. Adv Ther 2006; 23(1): 1-11.
- Stolk-Engelaar V, Meis J, Mulder J, Loeffen F, Hoogkamp-Korstanje J. Activity of 24 antimicrobials against Yersinia enterocolitica. Contrib Microbiol Immunol 1995; 13: 172-4.
- Bottone EJ. Yersinia enterocolitica: the charisma continues. Clin Microbiol Rev 1997; 10(2): 257-76.
- Pham JN, Bell SM, Hardy MJ, Martin L, Guiyoule A, Camiel E. Comparison of beta-lactamase production by Yersinia enterocolitica biotype 4, serotype O:3 isolated in eleven countries. Contrib Microbiol Immunol 1995; 13: 180-3.
- Frydén A, Bengtsson A, Foberg U, Svenungsson B, Castor B, Kärnell A, Schvarcz R, Lindblom B, Kihlström E. Early antibiotic treatment of reactive arthritis associated with enteric infections: clinical and serological study. BMJ 1990; 301(6764): 1299-302.
- Mansel JK, Rosenow EC 3rd, Smith TF, Martin JW Jr. Mycoplasma pneumoniae pneumonia. Chest 1989; 95(3): 639-46.
- Martin RE, Bates JH. Atypical pneumonia. Infect Dis Clin North Am 1991; 5(3): 585-601.
- Koskiniemi M. CNS manifestations associated with Mycoplasma pneumoniae infections: summary of cases at the University of Helsinki and review. Clin Infect Dis 1993; 17 Suppl 1: S52-7.
- Daxboeck F. Mycplasma pneumoniae central nervous system infections. Curr Opin Neurol 2006; 19(4): 374-8.
- Bitnun A, Ford-Jones E, Blaser S, Richardson S. Mycoplasma pneumoniae encephalitis. Semin Pediatr Infect Dis 2003; 14(2): 96-107.
- Smith R, Eviatar L. Neurologic manifestations of Mycoplasma pneumoniae infections: diverse spectrum of diseases. A report of six cases and review of the literature. Clin Pediatr (Phila) 2000; 39(4): 195-201.
- Tsiodras S, Kelesidis T, Kelesidis I, Voumbourakis K, Giamarellou H. Mycoplasma pneumoniae-associated myelitis: a comprehensive review. Eur J Neurol 2006; 13(2): 112-24.
- Chaudhry R, Nisar N, Malhotra P, Kumar A, Chauhan VS. Polymerase chain reaction confirmed Mycoplasma pneumoniae arthritis: a case report. Indian J Pathol Microbiol 2003; 46(3): 433-6.
- Vitullo BB, O’Regan S, de Chadarevian JP, Kaplan BS. Mycoplasma pneumonia associated with acute glomerulonephritis. Nephron 1978; 21(5): 284-8.
- Weinstein O, Shneck M, Levy J, Lifshitz T. Bilateral acute anterior uveitis as a presenting symptom of Mycoplasma pneumoniae infection. Can J Ophthalmol 2006; 41(5): 594-5.
- Yashar SS, Yashar B, Epstein E, Viani RM. Uveitis associated with Mycoplasma pneumoniae meningitis. Acta Ophthalmol Scand 2001; 79(1): 100-1.
- Ahvonen P, Sievers K, Aho K. Arthritis associated with Yersinia enterocolitica infection. Acta Rheumatol Scand 1969; 15(3): 232-53.
- Panush RS, Wallace DJ, Dorff RE, Engleman EP. Retraction of the suggestion to use the term “Reiter’s syndrome” sixty-five years later: the legacy of Reiter, a war criminal, should not be eponymic honor but rather condemnation. Arthritis Rheum 2007; 56(2): 693-4.
- Keynan Y, Rimar D. Reactive arthritis -- the appropriate name. Isr Med Assoc J 2008; 10(4): 256-8.
- Hannu T, Inman R, Granfors K, Leirisalo-Repo M. Reactive arthritis or post-infectious arthritis? Best Pract Res Clin Rheumatol 2006; 20(3): 419-33.
- Braun J, Kingsley G, van der Hejide D, Sieper J. On the difficulties of establishing a consensus on the definition of and diagnostic investigations for reactive arthritis. Results and discussion of a questionnaire prepared for the 4th International Workshop on Reactive Arthritis, Berlin, Germany, July 3-6, 1999. J Rheumatol 2000; 27(9): 2185-92.
- Leirisalo-Repo M. Reactive arthritis. Scand J Rheumatol 2005; 34(4): 251-9.
- Fendler C, Laitko S, Sörensen H, Gripenberg-Lerche C, Groh A, Uksila J, Granfors K, Braun J, Sieper J. Frequency of triggering bacteria in patients with reactive arthritis and undifferentiated oligoarthritis and the relative importance of the tests used for diagnosis. Ann Rheum Dis 2001; 60(4): 337-43.
- Leirisalo-Repo M. Sieper J Reactive arthritis: epidemiology, clinical features and treatment in ankylosing spondylitis and the spondyloarthropathies. MH Weisman, D van der Heijde and JD Reveille, Editors, Mosby Elevier: Philadelphia, p.53-64, 2006.
- Yu DT. Reactive arthritis (formerly Reiter syndrome). UpToDate, 2008.
- Clegg DO, Reda DJ, Weisman MH, Cush JJ, Vasey FB, Schumacher HR Jr, Budiman-Mak E, Balestra DJ, Blackburn WD, Cannon GW, Inman RD, Alepa FP, Mejias E, Cohen MR, Makkena R, Mahowald ML, Higashida J, Silverman SL, Parhami N, Buxbaum J, Haakenson CM, Ward RH, Manaster BJ, Anderson RJ, Henderson WG, et al. Comparison of sulfasalazine and placebo in the treatment of reactive arthritis (Reiter’s syndrome). A Department of Veterans Affairs Cooperative Study. Arthritis Rheum 1996; 39(12): 2021-7.
- Flagg SD, Meador R, Hsia E, Kitumnuaypong T, Schumacher HR Jr. Decreased pain and synovial inflammation after etanercept therapy in patients with reactive and undifferentiated arthritis: an open-label trial. Arthritis Rheum 2005; 53(4): 613-7.
- Kvien TK, Gaston JS, Bardin T, Butrimiene I, Dijkmans BA, Leirisalo-Repo M, Solakov P, Altwegg M, Mowinckel P, Plan PA, Vischer T; EULAR. Three month treatment of reactive arthritis with azithromycin: a EULAR double blind, placebo controlled study. Ann Rheum Dis 2004; 63(9): 1113-9.
- Yli-Kerttula T, Luukkainen R, Yli-Kerttula U, Möttönen T, Hakola M, Korpela M, Sanila M, Uksila J, Toivanen A. Effect of a three month course of ciprofloxacin on the late prognosis of reactive arthritis. Ann Rheum Dis 2003; 62(9): 880-4.
- Lauhio A, Leirisalo-Repo M, Lähdevirta J, Saikku P, Repo H. Double-blind, placebo-controlled study of three-month treatment with lymecycline in reactive arthritis, with special reference to Chlamydia arthritis. Arthritis Rheum 1991; 34(1): 6-14.
- Yli-Kerttula T, Luukkainen R, Yli-Kerttula U, Möttönen T, Hakola M, Korpela M, Sanila M, Parviainen J, Uksila J, Vainionpää R, Toivanen A. Effect of a three month course of ciprofloxacin on the outcome of reactive arthritis. Ann Rheum Dis 2000; 59(7): 565-70.
- Laasila N, Laasonen L, Leirisalo-Repo M. Antibiotic treatment and long term prognosis of reactive arthritis. Ann Rheum Dis 2003; 62(7): 655-8.
- Putschky N, Pott HG, Kuipers JG, Zeidler H, Hammer M, Wollenhaupt J. Comparing 10-day and 4-month doxycycline courses for treatment of Chlamydia trachomatis-reactive arthritis: a prospective, double-blind trial. Ann Rheum Dis, 2006; 65(11): 1521-4
- Fournier PE, Mainardi JL, Raoult D. Value of microimmunofluorescence for diagnosis and follow-up of Bartonella endocarditis. Clin Diagn Lab Immunol 2002; 9(4): 795-801.
- Kerkhoff FT, Rothova A. Bartonella henselae associated uveitis and HLA-B27. Br J Ophthalmol 2000; 84(10): 1125-9.
- Carter JD, Espinoza LR, Inman RD, Sneed KB, Ricca LR, Vasey FB, Valeriano J, Stanich JA, Oszust C, Gerard HC, Hudson AP. Combination antibiotics as a treatment for chronic Chlamydia-induced reactive arthritis: a double-blind, placebo-controlled, prospective trial. Arthritis Rheum 2010; 62(5): 1298-307.
- Gérard HC, Whittum-Hudson JA, Carter JD, Hudson AP. Molecular biology of infectious agents in chronic arthritis. Rheum Dis Clin North Am 2009; 35(1): 1-19.
- Carter JD, Gérard HC, Espinoza LR, Ricca LR, Valeriano J, Snelgrove J, Oszust C, Vasey FB, Hudson AP. Chlamydiae as etiologic agents in chronic undifferentiated spondylarthritis. Arthritis Rheum 2009; 60(5): 1311-6.
- Fainardi E, Castellazzi M, Tamborino C, Seraceni S, Tola MR, Granieri E, Contini C. Chlamydia pneumoniae-specific intrathecal oligoclonal antibody response is predominantly detected in a subset of multiple sclerosis patients with progressive forms. J Neurovirol 2009; 15(5-6): 425-33.
- Appelt DM, Roupas MR, Way DS, Bell MG, Albert EV, Hammond CJ, Balin BJ. Inhibition of apoptosis in neuronal cells infected with Chlamydophila (Chlamydia) pneumoniae. BMC Neurosci 2008; 9: 13.
- Contini C, Cultrera R, Seraceni S, Castellazzi M, Granieri E, Fainardi E. Cerebrospinal fluid molecular demonstration of Chlamydia pneumoniae DNA is associated to clinical and brain magnetic resonance imaging activity in a subset of patients with relapsing-remitting multiple sclerosis. Mult Scler 2004; 0(4): 360-9.
- Gerard HC, Wang Z, Whittum-Hudson JA, El-Gabalawy H, Goldbach-Mansky R, Bardin T, Schumacher HR, Hudson AP. Cytokine and chemokine mRNA produced in synovial tissue chronically infected with Chlamydia trachomatis and C. pneumoniae. J Rheumatol 2002; 29(9): 1827-35.
- De Backer J, Mak R, De Bacquer D, Van Renterghem L, Verbraekel E, Kornitzer M, De Backer G. Parameters of inflammation and infection in a community based case-control study of coronary heart disease. Atherosclerosis 2002; 160(2): 457-63.
- Appelbaum PC, Gillespie SH, Burley CJ, Tillotson GS. Antimicrobial selection for community-acquired lower respiratory tract infections in the 21st century: a review of gemifloxacin. Int J Antimicrob Agents 2004; 23(6): 533-46.
- Villareal C, Whittum-Hudson JA, Hudson AP. Persistent Chlamydiae and chronic arthritis. Arthritis Res 2002; 4(1): 5-9.
- Beutler AM, Hudson AP, Whittum-Hudson JA, Salameh WA, Gerard HC, Branigan PJ, Schumacher HR Jr. Chlamydia trachomatis Can Persist in Joint Tissue After Antibiotic Treatment in Chronic Reiter’s Syndrome / Reactive Arthritis. J Clin Rheumatol 1997; 3(3): 125-130.
- Leirisalo-Repo M. Are antibiotics of any use in reactive arthritis? APMIS 1993; 101(8): 575-81.
- Saebo A, Lassen J. Yersinia enterocolitica: an inducer of chronic inflammation. Int J Tissue React 1994; 16(2): 51-7.
- van der Heijden IM, Res PC, Wilbrink B, Leow A, Breedveld FC, Heesemann J, Tak PP. Yersinia enterocolitica: a cause of chronic polyarthritis. Clin Infect Dis 1997; 25(4): 831-7.
- Braun J, Tuszewski M, Eggens U, Mertz A, Schauer-Petrowskaja C, Döring E, Laitko S, Distler A, Sieper J, Ehlers S. Nested polymerase chain reaction strategy simultaneously targeting DNA sequences of multiple bacterial species in inflammatory joint diseases. I. Screening of synovial samples of patients with spondyloarthropathies and other arthritides. J Rheumatol 1997; 24(6): 1092-100.
- Makhnev MV. [The nature, frequency, specificity and duration of the retention of changes in different parts of the gastrointestinal tract in yersiniosis]. [Article in Russian].Ter Arkh 1994; 66(11): 12-7.
- Saebo A, Nyland H, Lassen J. Yersinia enterocolitica infection--an unrecognized cause of acute and chronic neurological disease? A 10-year follow-up study on 458 hospitalized patients. Med Hypotheses 1993; 41(3): 282-6.
- Borg AA, Gray J, Dawes PT. Yersinia-related arthritis in the United Kingdom. A report of 12 cases and review of the literature. Q J Med 1992; 84(304): 575-82.
- Saebo A, Lassen J. Acute and chronic pancreatic disease associated with Yersinia enterocolitica infection: a Norwegian 10-year follow-up study of 458 hospitalized patients. J Intern Med 1992; 231 (5): 537-41.
- Saebo A, Lassen J. Acute and chronic gastrointestinal manifestations associated with Yersinia enterocolitica infection. A Norwegian 10-year follow-up study on 458 hospitalized patients. Ann Surg 1992; 215(3): 250-5.
- Lindholm H, Visakorpi R. Late complications after a Yersinia enterocolitica epidemic: a follow up study. Ann Rheum Dis 1991; 50(10): 694-6.
- Saebo A, Lassen J. A survey of acute and chronic disease associated with Yersinia enterocolitica infection. A Norwegian 10-year follow-up study on 458 hospitalized patients. Scand J Infect Dis 1991; 23(5): 517-27.
- Fordham JN, Maitra S. Post-yersinial arthritis in Cleveland, England. Ann Rheum Dis 1989; 48(2): 139-42.
- Saebo A, Vik E, Lange OJ, Matuszkiewicz L. Inflammatory bowel disease associated with Yersinia enterocolitica O:3 infection. Eur J Intern Med 2005; 16(3): 176-182.
- Hoogkamp-Korstanje JA, de Koning J, Heesemann J. Persistence of Yersinia enterocolitica in man. Infection 1988; 16(2): 81-5.
- Fusco C, Bonini E, Soncini G, Frattini D, Giovannini S, Della Giustina E. Transient basal ganglia and thalamic involvement following Mycoplasma pneumoniae infection associated with antiganglioside antibodies. J Child Neurol 2010; 25(8): 1029-33.
- Hsing J, Welgampola M, Kieman MC. Reversible myeloradiculopathy due to Mycoplasma pneumoniae. J Clin Neurosci 2007; 14(1): 61-4.
- Gorthi SP, Kapoor L, Chaudhry R, Sharma N, Perez-Perez GI, Panigrahi P, Behari M. Guillain-Barré syndrome: association with Campylobacter jejuni and Mycoplasma pneumoniae infections in India. Natl Med J India 2006; 19(3): 137-9.
- Manteau C, Liest JM, Caillon J, M‘Guyen S, Quere MP, Roze JC, Gras-Le Guen C. Acute severe spinal cord dysfunction in a child with meningitis: Streptococcus pneumoniae and Mycoplasma pneumoniae co-infection. Acta Paediatr 2005; 94(9): 1339-41.
- Tsiodras S, Kelesidis I, Kelesidis T, Stamboulis E, Gimarellou H. Central nervous system manifestations of Mycoplasma pneumoniae infections. J Infect 2005; 51(5): 343-54.
- Trad S, Ghosn J, Dormont D, Stankoff B, Bricaire F, Caumes E. Nuclear bilateral Bell’s palsy and ageusia associated with Mycoplasma pneumoniae pulmonary infection. J Med Microbiol 2005; 54(Pt4): 417-9.
- Timitilli A, Di Rocco M, Nattero G, Tacchella A, Giacchino R. Unusual manifestations of infections due to Mycoplasma pneumoniae in children. Infez Med 2004; 12(2): 113-7.
- Völter C, Helms J, Weissbrich B, Rieckmann P, Abele-Horn M. Frequent detection of Mycoplasma pneumoniae in Bell’s palsy. Eur Arch Otorhinolaryngol 2004; 261(7): 400-4.
- Ashtekar CS, Jaspan T, Thomas D, Weston V, Gayatri NA, Whitehouse WB. Acute bilateral thalamic necrosis in a child with Mycoplasma pneumoniae. Dev Med Child Neurol 2003; 45(9): 634-7.
- Pfausler B, Engelhardt K, Kampfl A, Spiss H, Tafemer E, Schmutzhard E. Post-infectious central and peripheral nervous system diseases complicating Mycoplasma pneumoniae infection. Report of three cases and review of the literature. Eur J Neurol 2002; 9(1): 93-6.
- Goebels N, Helmchen C, Abele-Horn M, Gasser T, Pfister HW. Extensive myelitis associated with Mycoplasma pneumoniae infection: magnetic resonance imaging and clinical long-term follow-up. J Neurol 2001; 248(3): 204-8.
- Socan M, Ravnik I, Bencina D, Dovc P, Zakotnik B, Jazbec J. Neurological symptoms in patients whose cerebrospinal fluid is culture- and/or polymerase chain reaction-positive for Mycoplasma pneumoniae. Clin Infect Dis 2001; 32(2): E31-5.
- Rabay-Chacar H, Rizkallah E, Hakimeh NI, Khoury L, Merhej MT. Neurological complications associated with Mycoplasma pneumoniae infection. A case report. J Med Liban 2000; 48(2): 108-11.
- Van Koningsveld R, Van Doom PA, Schmitz PI, Ang CW, Van der Meché FG. Mild forms of Guillain-Barré syndrome in an epidemiologic survey in The Netherlands. Neurology 2000; 54(3): 620-5.
- Dionisio D, Valassina M, Mata S, Rossetti R, Vivarelli A, Esperti FC, Benvenuti M, Catalani C, Uberti M. Encephalitis caused directly by Mycoplasma pneumoniae. Scand J Infect Dis 1999; 31(5): 506-9.
- Abele-Horn M, Franck W, Busch U, Nitschko H, Roos R, Heesemann J. Transverse myelitis associated with Mycoplasma pneumoniae infection. Clin Infect Dis 1998; 26(4): 909-12.
- Narita M, Matsuzono Y, Itakura O, Togashi T, Kikuta H. Survey of mycoplasmal bacteremia detected in children by polymerase chain reaction. Clin Infect Dis 1996; 23(3): 522-5.
- Thomas NH, Collins JE, Robb SA, Robinson RO. Mycoplasma pneumoniae infection and neurological disease. Arch Dis Child 1993; 69(5): 573-6.
- Francis DA, Brown A, Miller DH, Wiles CM, Bennett ED, Leigh N. MRI appearances of the CNS manifestations of Mycoplasma pneumoniae: a report of two cases. J Neurol 1988; 235(7): 441-3.
- Carstensen H, Nilsson KO. Neurological complications associated with Mycoplasma pneumoniae infection in children. Neuropediatrics 1987; 18(1): 57-8.
- MacFarlane PI, Miller V. Transverse myelitis associated with Mycoplasma pneumoniae infection. Arch Dis Child 1984; 59(1): 80-2.
- Foy HM, Nolan CM, Allan ID. Epidemiologic aspects of M. pneumoniae disease complications: a review. Yale J Biol Med 1983; 56(5-6): 469-73.
- Cotter FE, Bainbridge D, Newland AC. Neurological deficit associated with Mycoplasma pneumoniae reversed by plasma exchange. Br Med J (Clin Res Ed) 1983; 286(6358): 22.
- Maida E, Kristoferitsch W. Cerebrospinal fluid findings in mycoplasma pneumoniae infections with neurological complications. Acta Neurol Scand 1982; 65(5): 524-38.
- Bayer AS, Galpin JE, Theofilopoulos AN, Guze LB. Neurological disease associated with Mycoplasma pneumoniae pneumonitis: demonstration of viable Mycoplasma pneumoniae in cerebrospinal fluid and blood by radioisotopic and immunofluorescent tissue culture techniques. Ann Intern Med 1981; 94(1): 15-20.
- Nicholson G. Transverse myelitis complicating Mycoplasma pneumoniae infection. Postgrad Med J 1977; 53(616): 86-7.
- Mardh PA, Ursing B, Lind K. Persistent cerebellar symptoms after infection with Mycoplasma pneumoniae. Scand J Infect Dis 1975; 7(2): 157-60.
- Hely MA, Williamson PM, Terenty TR. Neurological complications of Mycoplasma pneumoniae infection. Clin Exp Neurol 1984; 20: 153-60.
- Koskiniemi M. CNS manifestations associated with Mycoplasma pneumoniae infections: summary of cases at the University of Helsinki and review. Clin Infect Dis 1993; 17 Suppl 1: S52-7.
- Assaad F, Gispen R, Kleemola M, Syrucek L, Esteves K. Neurological diseases associated with viral and Mycoplasma pneumoniae infections. Bull World Health Organ 1980; 58(2): 297-311.
- Lind K, Zoffmann H, Larsen SO, Jessen O. Mycoplasma pneumoniae infection associated with affection of the central nervous system. Acta Med Scnad 1979; 205(4): 325-32.
- Fink CG, Sillis M, Read SJ, Butler L, Pike M. Neurological disease associated with Mycoplasma pneumoniae infection. PCR evidence against a direct invasive mechanism. Clin Mol Pathol 1995; 48(1): M51-4.
- Di Maria A, Ruberto G, Redaelli C, Gualtieri G. Anterior uveitis associated with Mycoplasma pneumoniae pneumonia: a case report. Acta Ophthalmol Scand 1999; 77(3): 349-50.
- Said MH, Layani MP, Colon S, Faraji G, Gilastre C, Cochat P. Mycoplasma pneumoniae-associated nephritis in children. Pediatr Nephrol 1999; 13(1): 39-44.
- Schonwald S, Gunjaca M, Kolacny-Babic L, Car V, Gosev M. Comparison of azithromycin and erythromycin in the treatment of atypical pneumonias. J Antimocrob Chemother 1990; 25 Suppl A: 123-6.
- File TM Jr, Segreti J, Dunbar L, Player R, Kohler R, Williams RR, Kojak C, Rubin A. A mulicenter, randomized study comparing the efficacy and safety of intravenous and/or oral levofloxacin versus ceftriaxone and/or cefuroxime axetil in treatment of adults with community-acquired pneumonia. Antimicrob Agents Chemother 1997; 41(9): 1965-72.
- Miklossy J, Kasas S, Zum AD, McCall S, Yu S, McGeer PL. Persisting atypical and cystic forms of Borrelia burgdorferi and local inflammation in Lyme neuroborreliosis. J Neuroinflammation 2008; 5: 40.
- Almeida OP, Lautenschlager NT. Dementia associated with infectious diseases. Int Psychogeriatr 2005; 17 Suppl 1: S65-77.
- McDonald AB. Alzheimer’s disease Braak Stage progressions: reexamined and redefined as Borrelia infection transmission through neural circuits. Med Hypotheses 2007; 68(5): 1059-64.
- Fallon BA, Levin ES, Schweitzer PJ, Hardesty D. Inflammation and central nervous system Lyme disease. Neurobiol Dis 2010; 37(3): 534-41.
- Friedman CR, Neimann J, Wegener HC, Tauxe RV. Epidemiology of Campylobacter jejuni infections in the United States and other industrialized nations. In: Campylobacter, 2nd edition, Nachamkin, I, Blaser, MJ (eds), AM Soc Microbiol, Washington DC 2000, p. 121.
- Moran AP, Upton ME. Factors affecting production of coccoid forms by Campylobacter jejuni on solid media during incubation. J Appl Bacteriol 1987; 62(6): 527-37.
- Rollins DM, Colwell RR. Viable but nonculturable stage of Campylobacter jejuni and its role in survival in the natural aquatic environment. Appl Environ Microbiol 1986; 52(3): 531-8.
- Wassenaar TM, Blaser MJ. Pathophyiology of Campylobacter jejuni inefctions of humans. Microbes Infect 1999; 1(12): 1023-33.
- Whitehouse CA, Balbo PB, Pesci EC, Cottle DL, Mirabito PM, Pickett CL. Campylobacter jejuni cytolethal distending toxin causes a G2-phase cell cycle block. Infect Immun 1998; 66(5): 1934-40.
- Pickett CL, Pesci EC, Cottle DL, Russell G, Erdam AN, Zeytin H. Prevalence of cytolethal distending toxin production in Campylobacter jejuni and relatedness of Campylobacter sp. cdtB gene. Infect Immun 1996; 64(6): 2070-8.
- Skirrow MB, Blaser MJ. Campylobacter jejuni. In: Infections of the gastrointestinal tract, 2nd ed, Blaser, MJ, Smith, PD, Ravdin, JI, et al. (Eds), Lippincott Williams and Wilkens, Philadelphia 2002, p. 719.
- Hannu T, Kauppi M, Tuomala M, Laaksonen I, Klemets P, Kuusi M. Reactive arthritis following an outbreak of Campylobacter jejuni infection. J Rheumatol 2004; 31(3): 528-30.
- Garg AX, Pope JE, Thiessen-Philbrook H, Clark WF, Ouimet J, Walkerton Health Study Investigators. Arthritis risk after acute bacterial gastroenteritis. Rheumatology (Oxford) 2008; 47(2): 200-4.
- Townes JM, Deodhar AA, Laines ES, Smith K, Krug HE, Barkhuizen A, Thompson ME, Cieslak PR, Sobel J. Reactive arthritis following culture-confirmed infections with bacterial enteric pathogens in Minnesota and Oregon: a population-based study. Ann Rheum Dis 2008; 67(12): 1689-96.
- Shenker BJ, Besack D, McKay T, Pankoski L, Zekavat A, Demuth DR. Induction of cell cycle arrest in lymphocytes by Actinobacillus actinomycetemcomitans cytolethal distending toxin requires three subunits for maximum activity. J Immunol 2005; 174(4): 2228-34.
- Sorvillo FJ, Lieb LE, Waterman SH. Incidence of campylobacteriosis among patients with AIDS in Los Angeles County. J Acquir Immune Defic Syndr 1991; 4(6): 598-602.
- Melamed I, Bujanover Y, Igra YS, Schwartz D, Zakuth V, Spirer Z. Campylobacter enteritis in normal and immunodeficient children. Am J Dis Child 1983; 137(8): 752-3.
- Taylor DE, Chang N. In vitro susceptibilities of Campylobacter jejuni and Campylobacter coli to azithromycin and erythromycin. Animicrob Agents Chemother 1991; 35(9): 1917-8.
- Gaunt PD, Piddock LJ. Ciprofloxacin resistant Campylobacter spp. in humans: an epidemiological and laboratory study. J Antimicrob Chemother 1996; 37(4): 747-57.
- Lariviere LA, Gaudreau CL, Turgeon FF. Susceptibility of clinical isolates of Campylobacter jejuni to twenty-five antimicrobila agents. J Antimicrob Chemother 1986; 18(6): 681-5.
- Bonetti B, Monaco S, Ferrari S, Tezzon F, Rizzuto N. Demyelinating polyradiculoneuritis following Coxiella burnetti infection (Q fever). Ital J Neurol Sci 1991; 12(4): 415-7.
- Walid MS, Ajjan M, Ulm AJ. Subacute Transverse myelitis with Lyme profile dissociation. Ger Med Sci 2008; 6: Doc04.
- Bernit E, Pouget J, Janbon F, Dutronc H, Martinez P, Brouqui P, Raoult D. Neurological involvement in acute Q fever: a report of 29 cases and review of the literature. Arch Intern Med 2002; 162(6): 693-700.
- Shaked Y, Samra Y. Q fever meningoencephalitis associated with bilateral abducens nerve paralysis, bilateral optic neuritis and abnormal cerebrospinal fluid findings. Infection 1989; 17(6): 394-5.
- Brooks RG, Licitra CM, Peacock MG. Encephalitis caused by Coxiella burnetii. Ann Neurol 1986; 20(1): 91-3.
- Kofteridis DP, Mazokopakis EE, Tselentis Y, Gikas A. Neurological complications of acute Q fever infection. Eur J Epidemiol 2004; 19(11): 1051-4.
- Biswas S, Raoult D, Rolain J-M. Molecular Characterization of Resistance to Macrolides in Bartonella henselae. Anticibrobial Agents and Chemotherapy 2006; 3192-93.
- Kordick DL, Papich MG, Breitschwerdt EB. Efficacy of Enrofloxacin or Doxycycline for Treatment of Bartonella henselae or Bartonella clarridgeiae Infection in Cats. Anticibrobial Agents and Chemotherapy 1997; 2448-55.
- Rolain JM, Brouqui P, Koehler JE, Maguina C, Dolan MJ, Raoult D. Minireview. Recommendations for Treatment of Human Infections Caused by Bartonella Species. Anticibrobial Agents and Chemotherapy 2004; 1921-33.
- Musso D, Drancourt M, Raoult D. Lack of bactericidal effect of antibiotics except aminoglycosides on Bartonella (Rochalimaea) henselae. The British Society for Antimicrobial Chemotherapy 1995.
- Pendle S, Ginn A, Iredell J. Antibicrobial susceptibility of Bartonella henselae using Etest methodology. Anticibrobial Agents and Chemotherapy 2006; 761-63.
- Dörbecker C, Sander A, Oberle K, Schülin-Casonato T. In vitro susceptivility of Bartonella species in 17 antimicrobial compounds: comparison of Etest and agar dilution. Anticibrobial Agents and Chemotherapy 2006; 784-88.
- Biswas S, Maggi RC, Papich MG, Keil D, Breitschwerdt EB. Comparative Activity of Pradofloxacin, Enrofloxacin, and Azithromycin against Bartonella henselae Isolates Collected from Cats and a Human. Journal of Clinical Microbiology 2010; 617-18.
- Florin TA, Zaoutis TE, Zaoutis LB. Beyond Cat Scratch Disease: Widening Spectrum of Bartonella henselae Infektion. Pediatrics 2008; 121;e1413.
- Tsuneoka H, Yanagihara M, Nojima J, Ichihara K. Anitmicrobial susceptibility by Etest of Bartonella henselae isolated from cats and human in Japan. In Infect Chemother 2010; 16(6):446-8
- Conrad DA. Treatment of cat-scratch disease. Curr Opin Pediatr 2001; 13(1):56-9.
- Rolain JM, Maurin M, Raoult D. Bactericidal effect of antibiotics on Bartonella and Brucella spp.: clinical implications. J Antimicrob Chemother 2000; 46(5):811-4.
- Ives TJ, Marston EL, Regnery RL, Butts JD, Majerus TC. In vitro susceptibilities of Rickettsia and Bartonella spp. to 14-hydroxy-clarithromycin as determined by immunofluorescent antibody analysis of infected vero cell monolayers. J Antimicrob Chemother 2000; 45(3):305-10.
- Kordick DL, Papich MG, Breitschwerdt EB. Efficacy of enrofloxacin or doxycycline for treatment of Bartonella henselae or Bartonella clarridgeiae infection in cats. Antimicrob Agents Chemother 1997; 41(11):2448-55.
- Maurin M, Birtles R, Raoult D. Current knowledge of Bartonella species. Eur J Clin Microbiol Infect Dis 1997; 16(7):487-506.
- Ives TJ, Manzewitsch P, Regnery RL, Butts JD, Kebede M. In vitro susceptibilities of Bartonella henselae, B. quintana, B. elizabethae, Rickettsia rickettsii, R. conorii, R. akari, and R. prowazekii to macrolide antibiotics as determined by immunofluorescent-antibody analysis of infected Vero cell monolayers. Antimicrob Agents Chemother 1997; 41(3):578-82.
- Musso D, Drancourt M, Raoult D. Lack of bactericidal effect of antibiotics except aminoglycosides on Bartonella (Rochalimaea) henselae. J Antimicrob Chemother 1995; 36(1):101-8.
- Sander A. Epidemiologie, Klinik und Diagnostik von Bartonella-Infektionen. Institut für Medizinische Mikrobiologie und Hygiene der Universität Freiburg. Antibiotika Monitor 5/03.
- Sobraqués M, Maurin M, Birtels RJ, Raoult D. In Vitro Susceptibilities of Four Bartonella bacilliformis Strains to 30 Antibiotic Compounds. Anticibrobial Agents and Chemotherapy 1999; 2090-92.
- von Baehr V. Die Labordiagnostik der Borrelieninfektion. umwelt-magazin-gesellschaft 2/2009; 22:119-124.
- Strauss B, Löschau M, Seidel T, Stallmach A, Thomas A. Are fatigue symptoms and chronic fatigue syndrome following Q fever infection related to psychosocial variables? J Psychosom Res 2012; 72(4):300-4.
- van der Hoek W, Schneeberger PM, Oomen T, Wegdam-Blans MC, Dijkstra F, Notermans DW, Bijlmer HA, Groeneveld K, Wijkmans CJ, Rietveld A, Kampschreur LM, van Duynhoven Y. Shifting priorities in the aftermath of a Q fever epidemic in 2007 to 2009 in The Netherlands: from acute to chronic infection. Euro Surveill 2012; 17(3):20059.
- Ledina D, Bradaric N, Miles I, Ivic I, Brncic N, Kuzmicic N. Chronic fatigue syndrome after Q fever. Med Sci Monit 2007; 13(7):CS88-92.
- Ayres JG, Flint N, Smith EG, Tunnicliffe WS, Fletcher TJ, Hammond K, Ward D, Marmion BP. Post-infection fatigue syndrome following Q fever. QJM 1998; 91(2):105-23.
- Brouqui P, Dupont HAT, Drancourt M, Berland Y, Etienne J, Leport C, Goldstein F, Massip P, Micoud M, Bertrand A, et al. Chronic Q fever. Ninety-two cases from France, including 27 cases without endocarditis. Arch Intern Med 1993; 153(5):642-8.
- Raoult D, Levy PY, Harlé JR, Etienne J, Massip P, Goldstein F, Micoud M, Beytout J, Gallais H, Remy G, et al. Chronic Q fever: diagnosis and follow-up. Ann N Y Acad Sci 1990; 590:51-60.
- Krauss H, Schmeer N, Schiefer HG. Epidemiology and significance of Q feber in the Federal Republic of Germany. Zentralbl Bakteriol Mikrobiol Hy A 1987; 267(1):41-50.
- Fenollar F, Fournier PE, Carrieri MP, Habib G, Messana T, Raoult D. Risks factors and prevention of Q fever endocarditis. Clin Infect Dis 2001; 33:312.
- Gikas A, Kofteridis DP, Manios A, Pediaditis J, Tselentis Y. Newer macrolides as empiric treatment for acute Q fever infection. Antimicrob Agents Chemother 2001; 45:3644.
- Limonard GJ, Peters JB, Nabuurs-Franssen MH, Weers-Pothoff G, Besselink R, Groot CA, Dekhuijzen PN, Vercoulen JH. Detailed analysis of health status of Q fever patients 1 year after the first Dutch outbreak: a case-control study. QJM 2010; 103(12):953-8.
- Wegdam-Blans et al., The Dutch Q fever Consensus Group, © 2011 The British Infection Association, published by Elsevier ltd.
- Lienhard R, Tritten ML, Siegrist HH, Gern L. First Case of Anaplasma phagocytophilum Seroconversion and Seroepidemiology in Northern Switzerland, 12th International Conference on Lyme Borreliosis and other tick-borne diseases, September 26-29, 2010, Ljubljana, Slovenia.
- Lommano E, Bertaiola L, Dupasquier C, Gern L. Prevalence of Rickettsia spp, Babesia spp and Anaplasma phagocytophilum in questing Ixodes ricinus ticks from different sites in Switzerland, 12th International Conference on Lyme Borreliosis and other tick-borne diseases, September 26-29, 2010, Ljubljana, Slovenia.
- Khanakah G, Leschnik M, Stanek G. Anaplasma, Borrelia, Coxiella, Rickettsia in ticks removed from dogs, 12th International Conference on Lyme Borreliosis and other tick-borne diseases, September 26-29, 2010, Ljubljana, Slovenia.
- Katargina O, Geller J, Vasilenko V, Golovljova I. Detection and Characterization of Babesia in Estonia, 12th International Conference on Lyme Borreliosis and other tick-borne diseases, September 26-29, 2010, Ljubljana, Slovenia.
- Breitschwerdt EB, Sontakke S, Hopkins S, Neurological manifestations of Bartonellosis in immunocompetent patients: A composite of reports from 2005-2012, Ashdin Publishing, Journal of Neuroparasitology Vol 3 (2012), Article ID 235640, 15 pages, doi:10.4303/jnp/235640.
- Publigheddu M, Giagheddu A, Genugu F, Giagheddu M, Marruso F, Epilepsia partialis continua in cat scratch disease, Seizure 2004; 13:191-195, doi:10.1016/S1059-1311(03)00159-6.
- Cotté V, Bonnet S, Le Rhun D, Le Naour E, Chauvin A, Boulouis HJ, Lecuelle B, Lilin T, Vayssier-Taussat M, Transmission of Bartonella henselae by Ixodes ricinus, Emerg Infect Dis 2008; 14(7):1074-80.
- Terford SR 3rd, Wormser GP, Bartonella spp. transmission by ticks not established, Emerg Infect Dis 2010; 16(3):379-84.
- Krause PJ, Spielman A, Telford SR 3rd, Sikand VK, McKay K, Christianson D, Pollack RJ, Brassard P, Magera J, Ryan R, Persing DH. Persistent parasitemia after acute babesiosis. N Engl J Med 1998; 339(3):160-5.
- Hatcher JC, Greenberg PD, Antique J, Jimenez-Lucho VE. Severe babesiosis in Long Island: review of 34 cases and their complications. Clin Infect Dis 2001; 32(8):1117-25.
- Hunfeld KP, Lambert A, Kampen H, Albert S, Epe C, Brade V, Tenter AM. Seroprevalence of Babesia infections in humans exposed to ticks in midwestern Germany. J Clin Microbiol 2002; 40(7):2431-6.
- Oleson CV, Sivalingam JJ, O’Neill BJ, Staas WE Jr. Transverse myelitis secondary to coexistent Lyme disease and babesiosis. J Spinal Cord Med 2003; 26(2):168-71.^
- Hildebrandt A, Hunfeld KP, Baier M, Krumbholz A, Sachse S, Lorenzen T, Kiehntopf M, Fricke HJ, Straube E. First confirmed autochthonous case of human Babesia microti infection in Europe. Eur J Clin Microbiol Infect Dis 2007; 26(8):595-601.
- Hunfeld KP, Hildebrandt A, Gray JS. Babesiosis: recent insights into an ancient disease. Int J Parasitol 2008; 38(11):1219-37.
- Ohmori S, Kawai A, Takada N, Saito-Ito A. Development of real-time PCR assay for differential detection and quantification for multiple Babesia microti-genotypes. Parasitol Int 2011; 60(4):403-9.
- Krause PJ. Babesiosis diagnosis and treatment. Vector Borne Zoonotic Dis 2003; 3(1):45-51.
- Häselbarth K, Tenter AM, Brade V, Krieger G, Hunfeld KP. First case of human babesiosis in Germany – Clinical presentation and molecular characterisation of the pathogen. Int J Med Microbiol 2007; 297(3):197-204.
- Wormser GP, Prasad A, Neuhaus E, Joshi S, Nowakowski J, Nelson J, Mittleman A, Aquero-Rosenfeld M, Topal J, Krause PJ. Emmergence of resistance to azithromycin-atovaquone in immunocompromised patients with Babesia microti infection. Clin Infect Dis 2010; 50(3):381-6.
- Hunfeld KP, Allwin R, Peters S, Kraiczy P, Brade V. Serologic evidence for tick-borne pathogens other than Borrelia burgdorferi (TOBB) in Lyme borreliosis patients from Midwesten Germany. Wien Klin Wochenschr 1998; 110/24:901-8.
- Harvey RA, Misselbeck WJ, Uphold RE. Cat-scratch disease: an unusual cause of combative Behavior. Am J Emerg Med. 1991; 9(1):52-3.
- Wheeler SW, Wolf SM, Steinberg EA. Cat-Scratch encephalopathy. Neurology 1997; 49(3):876-8.
- Seah AB, Azran MS, Rucker JC, Biousse V, Martin DF, Newman NJ. Magnetic resonance imaging abnormalties in cat-scratch diease encephalopathy. J Neuroophtalmol 2003; 23(1):16-21.
- Gerber JE, Johnson JE, Scott MA, Madhusudhan KT. Fatal meningitis and encephalitis due to Bartonella henselae bacteria. J Forensic Sci. 2002; 47(3):640-4.
- Hmaimess G, Kadhim H, Saint Matin C, Abu Serieh B, Mousny M, Sébire G. Cat scratch disease presenting as meningomyeloradiculopathy. Arch Dis Child. 2004; 89(7):691-2.
- Florin TA, Zaoutis TE, Zaoutis LB. Beyond cat scratch disease: widening spectrum of Bartonella henselae infection. Pediatrics. 2008; 121(5):e1413-25.
- Dreses-Werringloer U, Padubrin I, Zeidler H, Köhler L. Effects of Azithromycin and Rifampicin on Chlamydia trachomatis Infection In Vitro. Antimicrobial Agents and Chemotherapy 2001; 45(11):3001-3008.
- Freidank HM, Losch P, Vögele H, Wiedman-Al-Ahmad M. In Vitro Susceptibilities of Chlamydia pneumoniae Isolates from German Patients and Synergistic Activity of Antibiotic Combination. Antimicrobial Agents and Chemotherapy 1999; 43(7):1808-1801.